|
|
| | 1-broMo-4-phthaldehyde Basic information |
| Product Name: | 1-broMo-4-phthaldehyde | | Synonyms: | 1-BROMO-4-NAPHTHALDEHYDE;4-BROMO-1-NAPHTHALDEHYDE;4-BroMo-1-napthaldehyde;4-Bromo-1-naphthalenecarboxaldehyde;1-broMo-4-phthaldehyde;4-bromonaphthalene-1-carbaldehyde;1-Naphthalenecarboxaldehyde, 4-bromo-;4-bromo-1-naphaldehyde | | CAS: | 50672-84-9 | | MF: | C11H7BrO | | MW: | 235.08 | | EINECS: | | | Product Categories: | Miscellaneous Reagents | | Mol File: | 50672-84-9.mol | 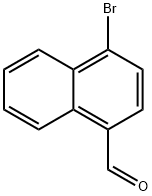 |
| | 1-broMo-4-phthaldehyde Chemical Properties |
| Melting point | 250℃ | | Boiling point | 359.4±25.0 °C(Predicted) | | density | 1.552±0.06 g/cm3(Predicted) | | storage temp. | under inert gas (nitrogen or Argon) at 2-8°C | | solubility | Chloroform (Slightly), Methanol (Slightly) | | form | Solid | | color | Off-White to Pale Beige |
| | 1-broMo-4-phthaldehyde Usage And Synthesis |
| Uses | 4-Bromo-1-napthaldehyde is used in the synthesis and antimicrobial activity of chalcones and flavones. | | Synthesis | 318 g of 1-bromo-4-bromomethylnaphthalene (II), 371.5 g of hexamethylenetetetramine, 795 g of glacial acetic acid, and 795 g of water were added to a 3L three-necked flask and reacted at reflux for 3 h at 100 °C. The reaction process was monitored by thin layer chromatography (TLC). Upon completion of the reaction, 636 g of concentrated hydrochloric acid was rapidly added dropwise for 0.5 hr. After the reaction mixture was cooled to room temperature, 4 L of water was added and stirred continuously for 3 hours. The solid product was collected by filtration, pulped with 2 L of water for 2 hours, filtered again, and the filter cake was washed twice with water, drained, and dried at 40 °C. The dried product was recrystallized with 318 g of ethanol and finally dried to give 200.4 g of 4-bromo-1-naphthaldehyde (III) in 80.4% yield. The molar ratio of 1-bromo-4-bromomethylnaphthalene (II) to hexamethylenetetetramine in the reaction was 1:2.5. | | References | [1] European Journal of Organic Chemistry, 2006, # 10, p. 2329 - 2335
[2] Patent: CN106366018, 2017, A. Location in patent: Paragraph 0040; 0041; 0042; 0043
[3] Inorganic Chemistry, 2016, vol. 55, # 5, p. 1986 - 1991
[4] Chemische Berichte, 1922, vol. 55, p. 1854
[5] Organic Letters, 2008, vol. 10, # 14, p. 3105 - 3108 |
| | 1-broMo-4-phthaldehyde Preparation Products And Raw materials |
|