|
|
| | TRIFLUOROSILANE Basic information |
| Product Name: | TRIFLUOROSILANE | | Synonyms: | TRIFLUOROSILANE;Trifluorosilaneintetrafluorosilane;TRIFLUOROSILANE: 20-25% SOLUTION IN TETRAFLUOROSILANE;20-25% SOLUTION IN TETRAFLUOROSILANE;TRIFLUOROSILANE (50-75%);SiHF3;Silane, trifluoro-(6CI,7CI,8CI,9CI);Trifluoro-(6Ci,7Ci,8Ci,9Ci)-Silane | | CAS: | 13465-71-9 | | MF: | F3HSi | | MW: | 86.09 | | EINECS: | | | Product Categories: | | | Mol File: | 13465-71-9.mol | 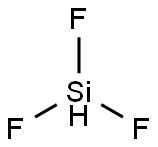 |
| | TRIFLUOROSILANE Chemical Properties |
| Melting point | -131°C | | Boiling point | -97,5°C | | density | 1.86 (0°C) [CRC10] | | form | colorless gas | | color | colorless | | Hydrolytic Sensitivity | 8: reacts rapidly with moisture, water, protic solvents |
| | TRIFLUOROSILANE Usage And Synthesis |
| Chemical Properties | colorless gas; enthalpy of vaporization 16.1kJ/mol; entropy of vaporization 87.9kJ/(mol·K) [CIC73] [CRC10] | | Synthesis | Trichlorosilane and TiF4 are heated for 18 hours in an autoclave onan oilbathat 100-200°C. If necessary, the reaction can be carried out in a sealed pressure tube. After cooling, the autoclave is slowly vented and the exit gases are collected in a quartz or glass trap cooled in liquid N2. The mixture is then fractionated. The residue in the autoclave consists of TiF4 and TiCl4. Since pure SiHF3 decomposes slowly even at room temperature it should be kept in Dry Ice or liquid N2. |
| | TRIFLUOROSILANE Preparation Products And Raw materials |
|