|
ChemicalBook Optimization Suppliers |
| 名前: |
BEST-REAGENT |
| 電話番号: |
400-1166-196 18981987031 |
| 電子メール: |
cdhxsj@163.com |
|
| | 2,2-ジメチル-3-(1,2,2,2-テトラブロモエチル)シクロプロパンカルボン酸シアノ(3-フェノキシフェニル)メチル 製品概要 |
| 化学名: | 2,2-ジメチル-3-(1,2,2,2-テトラブロモエチル)シクロプロパンカルボン酸シアノ(3-フェノキシフェニル)メチル | | 英語化学名: | TRALOMETHRIN | | 别名: | SCOUT;SCOUT(R);SAGA;SAGA(R);(S)-A-CYANO-3-PHENOXYBENZYL-(1R,3S)-2,2-DIMETHYL-3-[(RS)-1,2,2,2-TETRABROMOETHYL]-CYCLOPROPANE CARBOXYLATE;TRACKER;TRACKER(R);TRALATE | | CAS番号: | 66841-25-6 | | 分子式: | C22H19Br4NO3 | | 分子量: | 665.01 | | EINECS: | 266-493-1 | | カテゴリ情報: | Insecticide | | Mol File: | 66841-25-6.mol | 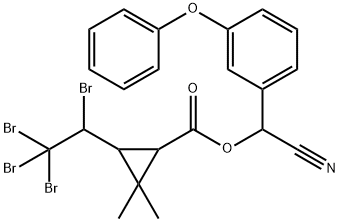 |
| | 2,2-ジメチル-3-(1,2,2,2-テトラブロモエチル)シクロプロパンカルボン酸シアノ(3-フェノキシフェニル)メチル 物理性質 |
| 融点 | 143℃ | | 沸点 | 185~190℃ (0.1mmHg) | | 比重(密度) | 1.70 g/cm3 (20℃) | | 蒸気圧 | 4.8×10-9 Pa (25 °C) | | 屈折率 | 1.6300 (estimate) | | 貯蔵温度 | 0-6°C | | 水溶解度 | 0.08 mg l-1 | | EPAの化学物質情報 | Tralomethrin (66841-25-6) |
| | 2,2-ジメチル-3-(1,2,2,2-テトラブロモエチル)シクロプロパンカルボン酸シアノ(3-フェノキシフェニル)メチル Usage And Synthesis |
| 外観 | ほとんど白色~黄赤色, 結晶性粉末又は塊 | | 溶解性 | アセトンに溶け、水にほとんど溶けない。水4.6mg/l(30℃), ベンゼン, キシレン500,アセトン, トルエン, トリクロロエタン400, シクロヘキサノン, 酢酸エチル,メチルナフタレン350, メチルイソブチルケトン250, メタノール53, ケロシン30,ヘキサン20(全てg/kg)。 | | 用途 | トラロメトリン試験用標準品。 | | 説明 | Orange-to-yellow resinous solid (technical grade
>39%) | | 使用 | Tralomethrin controls a range of insects, especially Lepidoptera, in
cereals, coffee, cotton, fruit, maize, oilseed rape, rice, tobacco and vegetables.
It is also effective for wood protection, household use, in public
health, in stored grains and for ectoparasite control on animals. | | 使用 | Tralomethrin can be used as insecticide. | | 定義 | ChEBI: A carboxylic ester resulting from the formal condensation between (1R)-cis-2,2-dimethyl-3-(1,2,2,2-tetrabromoethyl)cyclopropanecarboxylic acid and the alcoholic hydroxy group of (2S)-hydroxy(3-phenoxypheny
)acetonitrile. | | 农业用途 | Insecticide: Not approved for use in EU countries. Registered
for use in the U.S. | | 製品名 | DETHMOR®; HAG-107®; RU-25472®;
RU-25474®; SCOUT®; SCOUT® X-TRA Gel insecticide | | 代謝経路 | Tralomethrin probably owes most of its action to rapid conversion by
several mechanisms to deltamethrin. Information on its photodegradation,
degradation in soil, and its metabolism in plants, insects and animals
has been published and supports this conclusion. | | Toxicity evaluation | Acute oral LD50 for rats: 99-3,000 mg/kg depending on the
carrier used | | Degradation | Tralomethrin is stable as a solid but is labile to base. Acidic media reduce
the tendency to hydrolysis and epimerisation. It was readily debrominated
when solutions or thin films were irradiated at 360 nm or by
sunlight. Homolytic fission of a C-Br bond initiates this reaction,
which affords deltamethrin (2). Some epimerisation to trans-tralomethrin
and thence trans-deltamethrin also occurred. Dehydrobromination
also affords deltamethrin and the tribromo derivative (8). Ester cleavage
to the acid (3) and to the cyanohydrin (5) and thence to 3PBAl (6) and
3PBA (7) is important in the solid phase but not in solution (Ruzo and
Casida, 1981). The products are shown in Scheme 1. |
|