|
|
| | 4-(2H-1,2,3,4-TETRAAZOL-5-YL)BENZOIC ACID Basic information | | Uses |
| Product Name: | 4-(2H-1,2,3,4-TETRAAZOL-5-YL)BENZOIC ACID | | Synonyms: | BUTTPARK 21\02-63;4-(2H-TETRAZOL-5-YL)-BENZOIC ACID;4-(2H-1,2,3,4-TETRAAZOL-5-YL)BENZOIC ACID;TIMTEC-BB SBB002457;4(1H-tetrazd-5-yl)benzoic acid;5-(p-Carboxyphenyl)tetrazole;NSC 282009;4-(1H-Tetrazol-5-yl)benzoic acid 97% | | CAS: | 34114-12-0 | | MF: | C8H6N4O2 | | MW: | 190.16 | | EINECS: | | | Product Categories: | | | Mol File: | 34114-12-0.mol | 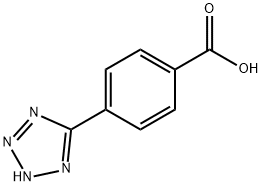 |
| | 4-(2H-1,2,3,4-TETRAAZOL-5-YL)BENZOIC ACID Chemical Properties |
| Melting point | 248-250℃ | | Boiling point | 467.5±47.0 °C(Predicted) | | density | 1.518 | | storage temp. | under inert gas (nitrogen or Argon) at 2-8°C | | pka | 3+-.0.10(Predicted) | | Appearance | Off-white to light yellow Solid | | InChI | InChI=1S/C8H6N4O2/c13-8(14)6-3-1-5(2-4-6)7-9-11-12-10-7/h1-4H,(H,13,14)(H,9,10,11,12) | | InChIKey | GEKBULKUEADYRB-UHFFFAOYSA-N | | SMILES | C(O)(=O)C1=CC=C(C2=NNN=N2)C=C1 |
| Hazard Codes | Xi,Xn | | Risk Statements | 22 | | HazardClass | IRRITANT |
| | 4-(2H-1,2,3,4-TETRAAZOL-5-YL)BENZOIC ACID Usage And Synthesis |
| Uses | 5-(4-Carboxyphenyl)-1H-tetrazole is an organic intermediate that can be prepared from 4-cyanobenzoic acid. Literature reports its potential use in the preparation of autocrine motor factor inhibitors. | | Synthesis | General procedure for the synthesis of 4-(1H-tetrazol-5-yl)benzoic acid from p-cyanobenzoic acid: p-cyanobenzoic acid (2 mmol), sodium azide (2.4 mmol, 1.2 eq.), and tetrabutylammonium bromide (2.4 mmol, 1.2 eq.) were added to a vial with a screw cap. The reaction mixture was stirred at 105 °C and the reaction progress was monitored by thin layer chromatography (TLC). Upon completion of the reaction, the mixture was cooled to room temperature and dissolved in deionized water (5 mL). Subsequently, the aqueous solution was acidified with 1 M hydrochloric acid to pH = 3. If precipitate formation was observed, the suspension was filtered and the filter cake was washed with deionized water to give pure 4-(1H-tetrazol-5-yl)benzoic acid. If no precipitate formed, the aqueous solution was extracted with ethyl acetate (3 x 4 mL). The organic phases were combined, washed with 1 M hydrochloric acid (3 x 4 mL), dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure to give pure 4-(1H-tetrazol-5-yl)benzoic acid. | | References | [1] Journal of Chemical Research, 2013, vol. 37, # 11, p. 665 - 667
[2] Angewandte Chemie - International Edition, 2018, vol. 57, # 2, p. 511 - 515
[3] Angew. Chem., 2018, vol. 130, # 2, p. 520 - 524,5
[4] European Journal of Organic Chemistry, 2014, vol. 2014, # 2, p. 436 - 441 |
| | 4-(2H-1,2,3,4-TETRAAZOL-5-YL)BENZOIC ACID Preparation Products And Raw materials |
|