|
|
| | xenytropium bromide Basic information |
| Product Name: | xenytropium bromide | | Synonyms: | xenytropium bromide;Xenytropium;[8-methyl-8-[(4-phenylphenyl)methyl]-8-azoniabicyclo[3.2.1]octan-3-yl] 3-hydroxy-2-phenylpropanoate bromide;[8-methyl-8-[(4-phenylphenyl)methyl]-8-azoniabicyclo[3.2.1]octan-3-yl] 3-hydroxy-2-phenyl-propanoate bromide;3-hydroxy-2-phenyl-propionic acid [8-methyl-8-(4-phenylbenzyl)-8-azoniabicyclo[3.2.1]octan-3-yl] ester bromide;N 399;N399;N-399 | | CAS: | 511-55-7 | | MF: | C30H34NO3.Br | | MW: | 536.507 | | EINECS: | 2081296 | | Product Categories: | | | Mol File: | 511-55-7.mol | 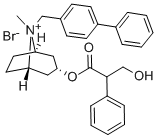 |
| | xenytropium bromide Chemical Properties |
| Toxicity | LD50 orl-rat: 1880 mg/kg OYYAA2 8,533,74 |
| | xenytropium bromide Usage And Synthesis |
| Originator | Xenytropium bromide,Licencia Budapest | | Manufacturing Process | 2 methods of preparation of endo-(+/-)-8-([1,1'-biphenyl]-4-ylmethyl)-3-(3-
hydroxy-1-oxo-2-phenylpropoxy)-8-methyl-8-azoniabicyclo[3.2.1]octane
bromide.
1). 28.9 g of dl-tropic acid ester of the tropine is dissolved in 300 ml of luke-
warm acetone and to the solution are added 25.0 g of 4-diphenyl-methyl-
bromide, dissolved in 75 ml of acetone. The solution is kept for 1 h at room
temperature, thereafter during 6 h at 40-60°C. The separated quaternary salt
is filtered off, washed with acetone and dried at gentle heat. The endo-(+/-)-
8-([1,1'-biphenyl]-4-ylmethyl)-3-(3-hydroxy-1-oxo-2-phenylpropoxy)-8-
methyl-8-azoniabicyclo[3.2.1]octane as snow-white microcrystalline powder,
melting point 220-222°C (dec., recystallizing from ethyl alcohol) is obtained
(yield 35.0-39.0 g).
2). 14.1 g of trans-tropanol are dissolved in 150 ml of acetone and the
solution mixed with 24.7 g of 4-diphenyl-methyl-bromide dissolved in a little
acetone. The solution is kept for some hours at about 50°C and therefater the
separated 4-diphenyl-methyl-trans-tropinium-bromide is isolated in the usual
manner. Yield about 90%. After recrystallising in ethyl alcohol the compound
melts at about 230°C (dec.).
19.4 g of the quarternary aminoalcohol obtained in the above manner is
mixed with 13.5 g of O-acetyl-dl-tropic acid bromide, in a vessel provided with
a tube containing calcium chloride, and the mixture is warmed in an oil bath
at 120-130°C until no more development of hydrogen bromide gas (approx. 3
h) takes place. For the purpose of splitting of the acetyl-residue the resulting
ester is boiled for 0.5 h with 50 ml of 10% hydrobromic acid and the resulting
solution evaporated to dryness in vacuo. On the endo-(+/-)-8-([1,1'-
biphenyl]-4-ylmethyl)-3-(3-hydroxy-1-oxo-2-phenylpropoxy)-8-methyl-8-
azoniabicyclo[3.2.1]octane, melting point 221°C (dec., recrystallization from
alcohol) is obtained. | | Therapeutic Function | Antimuscarinic, Spasmolytic | | Safety Profile | Poison by intravenous, subcutaneous, and intraperitoneal routes. Moderately toxic by ingestion. Experimental reproductive effects. When heated to decomposition it emits very toxic fumes of NOx and Br-. See also BROMIDES. |
| | xenytropium bromide Preparation Products And Raw materials |
|