| Company Name: |
Leancare Ltd.
|
| Tel: |
+33 962096793 |
| Email: |
enquiry@leancare.co.uk |
| Products Intro: |
|
|
| | METHYL GREEN Basic information |
| Product Name: | METHYL GREEN | | Synonyms: | PARIS GREEN;METHYL GREEN ZINC CHLORIDE DOUBLE SALT;BASIC BLUE 20;CI 42585;CI BASIC BLUE 20;CI NO 42590;CI NO 42585;COPPER (II) ACETOARSENITE | | CAS: | 54327-10-5 | | MF: | C26H33Cl2N3 | | MW: | 458.47 | | EINECS: | | | Product Categories: | Triphenylmethane | | Mol File: | 54327-10-5.mol | 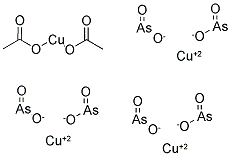 |
| | METHYL GREEN Chemical Properties |
| Melting point | >300°C | | solubility | Solubility Soluble in water, ethanol; insoluble in xylene | | form | powder | | color | Green crystalline | | PH Range | Yellow (0.1) to greenish-blue (2.3) | | λmax | 635nm, 420nm | | Major Application | Liquid crystal display device, photography, recording materials, inks, cosmetics, DNA staining, cancer detection |
| | METHYL GREEN Usage And Synthesis |
| Definition | ChEBI: Methyl green is an iminium salt composed of (4-{[4-(dimethylamino)phenyl][4-(trimethylazaniumyl)phenyl]methylidene}cyclohexa-2,5-dien-1-ylidene)(dimethyl)ammonium and chloride ions in a 1:2 ratio. A histological dye used to demonstrate nucleic acids by the Unna-Pappenheim stain, in conjunction with Pyronin Y. It has a role as a fluorochrome and a histological dye. It is an organic chloride salt, an iminium salt and a quaternary ammonium salt. It contains a methyl green(2+). | | Synthesis | Cupric oxide is heated with 8% acetic acid, As2O3 is added, and the mixture is refluxed for two hours. The product is allowed to cool for half an hour, filtered, washed and dried.Alternate method: Dilute acetic acid is allowed to react with an excess of freshly precipitated Cu(OH)2 and the product is separated by filtration. Dilute acetic acid is added to a solution of As2O3 in boiling NaOH until the color of phenolphthalein disappears. The hot solutions are mixed (mole ratio of CuO:As2O3 = 4:3) and allowed to stand for several days [S. Avery, J. Amer. Chem. Soc. 28, 1159 (1906)]. |
| | METHYL GREEN Preparation Products And Raw materials |
|