|
ChemicalBook Optimization Suppliers |
|
| 融点 | -76 °C | | 沸点 | 70 °C(lit.) | | 比重(密度) | 1.333 g/mL at 20 °C | | 蒸気圧 | <200 hPa (20 °C) | | 屈折率 | n20/D 1.500 | | 闪点 | 3 °F | | 貯蔵温度 | Store below +30°C. | | 溶解性 | sol chlorinated solvents and ethereal solvents; reacts
with protic solvents. | | 外見 | liquid | | 比重 | 1.0637 | | 色 | colorless | | 爆発限界(explosive limit) | 1.75-48.5%(V) | | 水溶解度 | reacts | | Sensitive | Moisture Sensitive | | Hydrolytic Sensitivity | 8: reacts rapidly with moisture, water, protic solvents | | BRN | 605287 | | 安定性: | Stable. Reacts violently with water and alcohols. Highly flammable. Incompatible with strong oxidizing agents, water, alcohols, caustics, ammonia. | | CAS データベース | 75-78-5(CAS DataBase Reference) | | NISTの化学物質情報 | Silane, dichlorodimethyl-(75-78-5) | | EPAの化学物質情報 | Dimethyldichlorosilane (75-78-5) |
| | ジクロロジメチルシラン Usage And Synthesis |
| 外観 | 無色~うすい黄色~うすい黄赤色透明液体 | | 解説 | ジメチルジクロロシラン,じめちるじくろろしらん,有機ケイ素化合物の一つ。常温で無色の液体。水に対してきわめて不安定であり、容易に反応して塩化水素を発生するため、刺激臭がある。 製法は、金属ケイ素と塩化メチルを触媒存在下で高温で反応させると他のメチルシラン類とともに得ることができ、蒸留で精製する。ポリジメチルシロキサンHO(SiMe2OSiMe2)nOHはこの物質が原料であり、条件により重合度を変えることができるので、工業的に非常に有用な原料である。合成化学的にはジオールの保護のためのシリル化剤として有用である。 | | 用途 | 主としてシラノールを経てポリシロキサンとして利用される。シリコーンゴム、シリコーンシーラント、シリコーン油およびケイ素樹脂の汎用グレードの主原料として利用されるほか、アルキド、エポキシ、アクリル等の樹脂、ポリカーボネート、ポリエーテル等の改質に用いられる。また各種のジメチルシリル基を含む化合物の合成原料とししても用いられる。 | | 説明 | Dimethyldichlorosilane is a colorless liquidwith sharp, irritating odor. Molecular weight= 129.07;Specific gravity = 1.07 at 25℃; Boiling point = 70.5℃;Melting point=86℃; Vapor pressure= 110 mmHg at20℃; Flash point = 27℃; Autoignition temperature=375℃. Explosive limits: LEL = 3.4%; UEL=9.5℃.Hazard Identification (based on NFPA-704 M Rating | | 化学的特性 | Colorless to brown liquid | | 物理的性質 | mp ?76°C; bp 70–71°C; d 1.064 g cm?3. | | 使用 | Dichlorodimethylsilane is used to prepare a resin bound siloxane with tertiary alcohols and it is also used as a reagent for synthesis of optically active ansa-mettallocene polymerization catalysts. It acts as a precursor to silicone and polysilane compounds. It is also used in the glass coating to protect it from micro particles. It is involved in the preparation of resin bound siloxane with reactivity towards tertiary alcohols. | | 使用 | Dichlorodimethylsilane can be used as additive for pinacol cyclization; protecting group for diols and
carbonyl compounds;precursor for a wide variety of siliconbased
reagents.
Dichlorodimethylsilane (1) allows clean
pinacol cyclization of a keto aldehyde to occur without competition
from an aldol reaction (eq 1).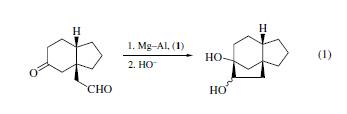 | | 使用 | Dichlorodimethylsilicon is a organosilicon compound and is the precursor to dimethylsilicone and polysilane compounds. | | 調製方法 | Produced by the action of silicon on methyl chloride in
presence of copper catalyst, or by Grignard reaction from
methyl chloride and silicon tetrachloride. | | 一般的な説明 | A colorless fuming liquid with a pungent odor. Flash point 16°F. Vapor and liquid may cause burns. Denser than water. Vapors heavier than air. | | 反応プロフィール | Chlorosilanes, such as Dichlorodimethylsilane, are compounds in which silicon is bonded to from one to four chlorine atoms with other bonds to hydrogen and/or alkyl groups. Chlorosilanes react with water, moist air, or steam to produce heat and toxic, corrosive fumes of hydrogen chloride. They may also produce flammable gaseous H2. They can serve as chlorination agents. Chlorosilanes react vigorously with both organic and inorganic acids and with bases to generate toxic or flammable gases. | | 健康ハザード | Inhalation irritates mucous membranes. Severe gastrointestinal damage may occur. Vapors cause severe eye and lung injury. Upon short contact, second and third degree burns may occur. | | 火災危険 | Vapor may explode if ignited in an enclosed area. Reacts vigorously with water to generate hydrogen chloride. Hydrogen chloride and phosgene gases may be formed upon heating or in fire. Runoff to sewer may create fire or explosion hazard. | | 安全性プロファイル | Poison by ingestion and
intraperitoneal routes. Moderately toxic by
inhalation. A skin and severe eye irritant.
Violent reaction on contact with water.
When heated to decomposition it emits
toxic fumes of Cl-. See also
CHLOROSILANES. | | 職業ばく露 | PrimaryIrritant. Used as an intermediate in the manufacture of silicone polymers. | | 応急処置 | If this chemical gets into the eyes, remove anycontact lenses at once and irrigate immediately for at least15 min, occasionally lifting upper and lower lids. Seek medical attention immediately. If this chemical contacts theskin, remove contaminated clothing and wash immediatelywith soap and water. Seek medical attention immediately. Ifthis chemical has been inhaled, remove from exposure,begin rescue breathing (using universal precautions, including resuscitation mask) if breathing has stopped and CPR ifheart action has stopped. Transfer promptly to a medicalfacility. When this chemical has been swallowed, get medical attention. If victim is conscious, administer water ormilk. Do not induce vomiting. Medical observation isrecommended for 2448 h after breathing overexposure, aspulmonary edema may be delayed. As first aid for pulmonary edema, a doctor or authorized paramedic may consideradministering a corticosteroid spray. | | 貯蔵 | (1) Color Code—Red: Flammability Hazard: Storein a flammable liquid storage area or approved cabinetaway from ignition sources and corrosive and reactivematerials. (2) Color Code—Blue: Health Hazard/Poison:Store in a secure poison location. Dimethyldichlorosilanemust be stored to avoid contact with oxidizers (such as perchlorates, peroxides, permanganates, chlorates, andnitrates), since violent reactions occur. Before entering confined space where this chemical may be present, check tomake sure that an explosive concentration does not exist.Store in tightly closed containers in a cool, well-ventilatedarea away from water, steam, or moisture, because toxicand corrosive hydrogen chloride gas can be produced. Donot store at temperatures above 50℃/122°F. Sources ofignition, such as smoking and open flames, are prohibitedwhere dimethyldichlorosilane is handled, used, or stored.Metal containers involving the transfer of 5 gallons or moreof dimethyldichlorosilane should be grounded and bonded. | | 輸送方法 | Dimethyldichlorosilane requires a shipping labelof “FLAMMABLE LIQUID, CORROSIVE.” It falls inHazard Class 3 and Packing Group II | | 純化方法 | Other impurities are chlorinated silanes and methylsilanes. Fractionate it through a 3/8in diameter 7ft Stedman column (p 11) rated at 100 theoretical plates at almost total reflux. See purification of MeSiCl2. Solutions in heptane, 1,1,1-trichloroethane or 1-chloronaphthalene are used for the silanization of glassware and pipettes. [Sauer & Hadsell J Am Chem Soc 70 3590 1948, Beilstein 4 IV 4110.] | | 不和合性 | Forms explosive gas mixture with air.Water, steam, and moisture forms toxic and corrosivehydrogen chloride gas. Incompatible with acetone, amines,ammonia, alcohols, strong oxidizers, caustics. Attacks mostmetals. Do not store in temperatures above 122°F/50℃. |
|