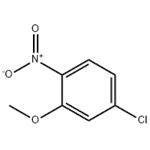
- 5-CHLORO-2-NITROANISOLE
-
- $100.00 / 1KG
-
2025-09-25
- CAS:6627-53-8
- Min. Order: 1KG
- Purity: 99%
- Supply Ability: g-kg-tons, free sample is available
- 5-Chloro-2-nitroanisole
-

- $0.00 / 1Kg
-
2020-02-26
- CAS: 6627-53-8
- Min. Order: 1KG
- Purity: 99.0%
- Supply Ability: 100 tons
|
| | 5-CHLORO-2-NITROANISOLE Basic information |
| | 5-CHLORO-2-NITROANISOLE Chemical Properties |
| Melting point | 70-72 °C(lit.) | | Boiling point | 289.1±20.0 °C(Predicted) | | density | 1.4219 (rough estimate) | | refractive index | 1.6000 (estimate) | | storage temp. | Sealed in dry,Room Temperature | | solubility | Chloroform, Ethyl Acetate | | form | Solid | | color | Yellow | | InChI | InChI=1S/C7H6ClNO3/c1-12-7-4-5(8)2-3-6(7)9(10)11/h2-4H,1H3 | | InChIKey | ABEUJUYEUCCZQF-UHFFFAOYSA-N | | SMILES | C1([N+]([O-])=O)=CC=C(Cl)C=C1OC | | CAS DataBase Reference | 6627-53-8(CAS DataBase Reference) | | EPA Substance Registry System | 5-Chloro-2-nitroanisole (6627-53-8) |
| Hazard Codes | Xi | | Risk Statements | 36/37/38 | | Safety Statements | 26-37/39-36 | | WGK Germany | 3 | | TSCA | TSCA listed | | HS Code | 2909309090 | | Storage Class | 6.1C - Combustible acute toxic Cat.3
toxic compounds or compounds which causing chronic effects | | Hazard Classifications | Acute Tox. 4 Oral
Carc. 1B Inhalation |
| | 5-CHLORO-2-NITROANISOLE Usage And Synthesis |
| Chemical Properties | SLIGHTLY YELLOW TO GREEN CRYSTALLINE POWDER | | Uses | 5-Chloro-2-nitroanisole is involved in the synthesis of orally bioavailable anaplastic lymphoma kinases (ALK) inhibitors as anticancer drugs (1). In addition, it is used in the synthesis of kinesin spindle protein (KSP) inhibitors which are responsible for the spindle pole separation which occurs during mitosis in cancer cells (2). | | Synthesis | To 5-chloro-2-nitrophenol (40 g, 0.23 mol) and iodomethane (49 g, 0.345 mol) in DMF (200 mL), K2CO3 (47.6 g, 0.345 mol) was added. The reaction mixture was stirred at room temperature for 16 hours. After completion of the reaction, the mixture was partitioned between ethyl acetate and water. The organic layer was separated, washed with brine, dried over MgSO4, filtered and concentrated in vacuum. The residue was purified by silica gel column chromatography (with petroleum ether as eluent) to afford the target product 2-nitro-5-chloroanisole (30 g, 70% yield). | | References | [1] Liebigs Annalen der Chemie, 1988, p. 203 - 208
[2] Patent: US2014/288045, 2014, A1. Location in patent: Paragraph 0461
[3] Patent: WO2016/44772, 2016, A1. Location in patent: Paragraph 323 |
| | 5-CHLORO-2-NITROANISOLE Preparation Products And Raw materials |
|