| Company Name: |
Shanghai Hongye Biotechnology Co. Ltd
|
| Tel: |
400-9205774 |
| Email: |
sales@glpbio.cn |
| Products Intro: |
Product Name:15-deoxy-δ12,14-Prostaglandin J2 Glutathione
CAS:537695-15-1
Purity:>98% Package:1mg;10mg;50mg;100mg;
|
| Company Name: |
ChemeGen(Shanghai) Biotechnology Co.,Ltd.
|
| Tel: |
18818260767 |
| Email: |
sales@chemegen.com |
| Products Intro: |
Product Name:15-deoxy-Δ12,14-Prostaglandin J2 Glutathione
CAS:537695-15-1
Purity:98% Package:10 mg;50 mg;100 mg;500 mg;1 g;5 g;10 g
|
| Company Name: |
Shanghai Yifei Biotechnology Co. , Ltd.
|
| Tel: |
021-65675885 18964387627 |
| Email: |
customer_service@efebio.com |
| Products Intro: |
Product Name:15-deoxy-Δ12,14-Prostaglandin J2 Glutathione
CAS:537695-15-1
Purity:90% Package:50μg;100μg;500μg
|
| Company Name: |
Cayman Chemical Company
|
| Tel: |
(800) 364-9897 |
| Email: |
cayman@caymanchem.com |
| Products Intro: |
Product Name:15-deoxy-Δ12,14-Prostaglandin J2 Glutathione
CAS:537695-15-1
|
|
| | 15-deoxy-Δ12,14-Prostaglandin J2 Glutathione Basic information |
| Product Name: | 15-deoxy-Δ12,14-Prostaglandin J2 Glutathione | | Synonyms: | 15-deoxy-Δ12,14-Prostaglandin J2 Glutathione;PJVSKAKTGJJITI-YPYHUWNQSA-N;Glycine, L-γ-glutamyl-S-[(1S,2R,3E)-2-[(2Z)-6-carboxy-2-hexen-1-yl]-3-(2Z)-2-octen-1-ylidene-4-oxocyclopentyl]-L-cysteinyl- | | CAS: | 537695-15-1 | | MF: | C30H45N3O9S | | MW: | 623.758 | | EINECS: | | | Product Categories: | | | Mol File: | 537695-15-1.mol | 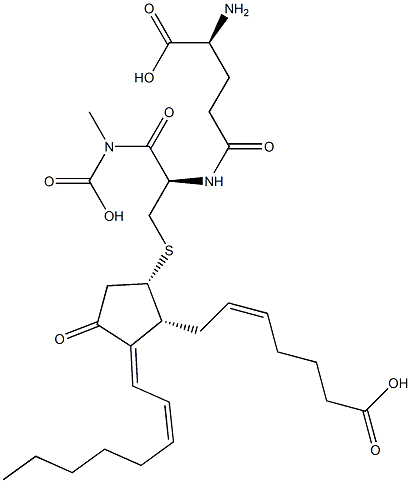 |
| | 15-deoxy-Δ12,14-Prostaglandin J2 Glutathione Chemical Properties |
| Boiling point | 963.6±65.0 °C(Predicted) | | density | 1.28±0.1 g/cm3(Predicted) | | solubility | DMF: 20 mg/ml; DMSO: 20 mg/ml; PBS (pH 7.2): 0.2 mg/ml | | pka | 2.21±0.10(Predicted) |
| | 15-deoxy-Δ12,14-Prostaglandin J2 Glutathione Usage And Synthesis |
| Description | 15-deoxy-Δ12,14-Prostaglandin J2 Glutathione (15-deoxy-Δ12,14-PGJ2 Glutathione) is a non-enzymatic adduct formed from 15-deoxy-Δ12,14-PGJ2 and glutathione.1,2,3 The biological properties of this compound have not been characterized. | | Definition | ChEBI: 15-deoxy-goyazensolide is a terpene lactone. | | References | 1. Cox, B., Murphey, L.J., Zackert, W.E., et al. Human colorectal cancer cells efficiently conjugate the cyclopentenone prostaglandin, prostaglandin J2, to glutathione Biochim. Biophys. Acta 1584,37-45(2002).
2. Brunoldi, E.M., Zanoni, G., Vidari, G., et al. Cyclopentenone prostaglandin, 15-deoxy-Δ12,14-PGJ2, is metabolized by HepG2 cells via conjugation with glutathione Chem. Res. Toxicol. 20,1528-1535(2007).
3. Paumi, C.M., Smitherman, P.K., Townsend, A.J., et al. Glutathione S-transferases (GSTs) inhibit transcriptional activation by the peroxisomal proliferator-activated receptor γ (PPARγ) ligand, 15-deoxy-Δ12,14 prostaglandin J2 (15-d-PGJ2). Biochemistry 43(8),2345-2352(2004). |
| | 15-deoxy-Δ12,14-Prostaglandin J2 Glutathione Preparation Products And Raw materials |
|