|
ChemicalBook Optimization Suppliers |
|
| | 3-(4-クロロフェニル)-1-メトキシ-1-メチル尿素 製品概要 |
| | 3-(4-クロロフェニル)-1-メトキシ-1-メチル尿素 物理性質 |
| 融点 | 81.5℃ | | 比重(密度) | 1.3575 (rough estimate) | | 屈折率 | 1.5790 (estimate) | | 闪点 | 100 °C | | 貯蔵温度 | Sealed in dry,Room Temperature | | 酸解離定数(Pka) | 12.80±0.70(Predicted) | | 色 | White to Light yellow to Light orange | | 水溶解度 | 0.58g/L(room temperature) | | BRN | 2212523 | | InChI | InChI=1S/C9H11ClN2O2/c1-12(14-2)9(13)11-8-5-3-7(10)4-6-8/h3-6H,1-2H3,(H,11,13) | | InChIKey | LKJPSUCKSLORMF-UHFFFAOYSA-N | | SMILES | N(OC)(C)C(NC1=CC=C(Cl)C=C1)=O | | EPAの化学物質情報 | Monolinuron (1746-81-2) |
| | 3-(4-クロロフェニル)-1-メトキシ-1-メチル尿素 Usage And Synthesis |
| 外観 | 白色, 結晶性粉末~粉末又は塊 | | 溶解性 | エタノール及びアセトンに溶け、水にほとんど溶けない。 | | 農薬用途 | 除草剤 | | 定義 | ChEBI: Monolinuron is a member of ureas. | | 反応性 |
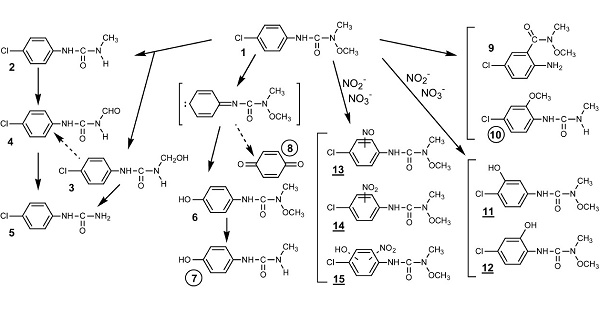
Nitrite or nitrate induced-photolysis of monolinuron (a phenyl urea herbicide) using the 300–450 nm light excitation gives rise to various photo-products. The direct photolysis of monolinuron proceeds via two main pathways yielding, respectively, 3-(4-chlorophenyl)-1-methylurea, which results from demethoxylation of the N-terminus substituted functional group and 3-(4-hydroxyphenyl)-1-methoxy-1- methylurea obtained by photohydrolysis of the C-Cl bond[1].
Degradation pathways of monolinuron by direct and nitrite (or nitrate) induced photolysis. Specific photoproducts: circled numbers (direct); underlined numbers (induced):
| | 安全性プロファイル | Moderately toxic by
ingestion. Experimental teratogenic and
reproductive effects. When heated to
decomposition it emits very toxic fumes of
Cland NOx. | | 参考文献 |
[1] S. Nélieu. “Nitrite and nitrate induced photodegradation of monolinuron in aqueous solution.” Environmental Chemistry Letters 2 2 (2004): 83–87.
|
|