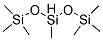Application of Poly(methylhydrosiloxane)
Aug 14,2019
Description
Poly(methylhydrosiloxane) is a reducing agent. For example, it can be used for the reduction of esters to alcohols as well as the reduction of aldehydes and ketone. It can also be used for the reduction of phosphine oxides to phosphine. Recent study has also used it for conjugate reduction of α, β-Unsaturated Carbonyl and Carboxyl Compounds. Finally, it is also used in greener amine synthesis mediated by reductive amination which is an alternative to borohydrides.
Application
Polymethylhydrosiloxane(PMHS) is an easily handled,inexpensive,non-toxic,and mild reducing agent. PMHS is attractive as a substitute for more expensive or hazardous silanes or siloxanes and as the stoichiometric reductant in catalytic organotin-mediated processes. It can be cross linked by metal catalyst at low temperature to form waterproof membrane. It is used as waterproof agent of textile, glass, pottery, paper, leather, metal, cement and marble etc, especially in textile waterproof. It is used as insulator and cross linker of paper. Poly(methylhydrosiloxane) is used in the reduction of esters to alcohols, catalyzed by a combination of titanocene dichloride and either n-BuLi or EtMgBr. It is a safer alternative to triethoxysilane, B22063, for reduction of phosphine oxides to phosphines, catalyzed by Ti(O-i-Pr)4. It is also used in greener amine synthesis by reductive amination as an alternative to borohydrides.
Preparative Methods
hydrolysis of methyldichlorosilane followed by heating (60–150 ◦C) the resultant mixture of cyclicsilanes in the presence of hexamethyldisiloxane generates the linear polysiloxane.
Handling, Storage, and Precautions
stable to air and moisture; incompatible with strong acids, bases, or oxidants (forms hydrogen upon decomposition); generally considered non-toxic, however thorough toxicity studies have not been performed; skin/eye contact and inhalation should be avoided.
Conclusion
Polymethylhydrosiloxane (PMHS) is an easily handled, inexpensive, non-toxic, and mild reducing agent. Although relatively inert towards organic functionality, PMHS can transfer its hydride to a variety of metal catalysts (including Sn, Ti, Zn, Cu, and Pd) which can then participate in a wide range of reductions. Alternatively, when made hypercoordinate by the action of fluoride or other nucleophiles, PMHS can act directly as a reducing agent. PMHS is attractive as a substitute for more expensive or hazardous silanes or siloxanes and as the stoichiometric reductant in catalytic organotin-mediated processes.
- Related articles
- Related Qustion
- Poly(methylhydrosiloxane): Overview and Conversion Method of its Waste to Useful Commodities Nov 5, 2024
Depolymerization of Poly(methylhydrosiloxane) waste with BF3OEt2 yields valuable MeSiF3 and MeSiF2H, vital for silicone industry, enhancing sustainability.
- Poly(methylhydrosiloxane): A Versatile Silicone Oil with Applications in Chemical Synthesis Dec 27, 2023
Poly(methylhydrosiloxane) is a versatile silicone oil used in waterproof coatings, organic synthesis, catalysis, and protective applications in biomedical and electronic fields.
- Exploring the versatile applications of poly(methylhydrosiloxane) in material engineering Jun 9, 2023
Poly(methylhydrosiloxane) is a versatile material that has numerous applications in the field of material engineering.
Sorafenib is used to treat kidney, liver, and thyroid cancer. It is a chemotherapy drug that works by slowing or stopping the growth of cancer cells.....
Aug 14,2019API1-Fluornaphthalene is an organofluorine chemical compound from the group of naphthalene derivatives and fluoroaromatics. It is a colorless, combustible liquid, which is insoluble in water.....
Aug 14,2019Organic ChemistryPoly(methylhydrosiloxane)
63148-57-2You may like
Poly(methylhydrosiloxane) manufacturers
- Poly(methylhydrosiloxane)
-

- 2025-12-05
- CAS:63148-57-2
- Min. Order:
- Purity: 0.99
- Supply Ability:
- Poly(methylhydrosiloxane)
-

- $10.00 / 1ASSAYS
- 2025-12-03
- CAS:63148-57-2
- Min. Order: 1ASSAYS
- Purity: 99%
- Supply Ability: 100 mt
- Poly(methylhydrosiloxane)
-
- $0.00 / 25KG
- 2025-12-01
- CAS:63148-57-2
- Min. Order: 1KG
- Purity: 98
- Supply Ability: 10000KGS