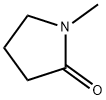
N-Methyl-2-pyrrolidone: Application, absorption, metabolism and toxicity
Apr 11,2023
General description
N-Methyl-2-pyrrolidone is an organic compound consisting of a 5-membered lactam. It is a colorless liquid, although impure samples can appear yellow. It is miscible with water and with most common organic solvents. It also belongs to the class of dipolar aprotic solvents such as dimethylformamide and dimethyl sulfoxide. It is used in the petrochemical and plastics industries as a solvent, exploiting its nonvolatility and ability to dissolve diverse materials [1]. Its appearance is as follows:

Figure 1 Appearance of N-Methyl-2-pyrrolidone.
Application
N-Methyl-2-pyrrolidone has been used in different fields of industrial applications, including electronics, petroleum, paint, textiles, rubber, chemical, polymer, and pharmaceutical industries; various syntheses; and different analytical methods [2]. N-Methyl-2-pyrrolidone is one of the main pharmaceutical cosolvents, and it acts as a very strong solubilizing agent; it is a solubilizing excipient used in parenteral and oral medications [3]. It is an important solvent used in the extraction, purification, and crystallization of drugs. N-Methyl-2-pyrrolidone can be used in parenteral formulations of drugs, because in addition to its excellent solubilizing power, it has low viscosity, which is a critical parameter in using fine-gauge needles or microcatheters [3]. In pharmaceutical applications, N-Methyl-2-pyrrolidone can also be used as a solubilizing agent for increasing the uptake of poorly soluble drugs such as itraconazole. Moreover, N-Methyl-2-pyrrolidone is a chemical penetration enhancer and is used for enhancement of transdermal delivery of hydrophilic and hydrophobic drugs from an aqueous phase [4]. It is used as a good enhancer in topical formulations of griseofulvin, delivering effective concentrations of the drug.
Absorption and metabolism
N-Methyl-2-pyrrolidone is easily absorbed from the human skin gastrointestinal and respiratory tracts. The permeability of commercial solvents through human skin has been investigated in a study, and the permeability rate of N-Methyl-2-pyrrolidone was found to be higher than that of other solvents [5]. It is quickly distributed in most organs, with a relatively high concentration in the sexual organs. Repeated exposures may be one of the reasons for infertility [6]. The volume of distribution (Vd) of N-Methyl-2-pyrrolidone is 0.7 L/kg, and the half-life of unchanged N-Methyl-2-pyrrolidone in the plasma after oral or dermal administration and inhalation exposure are 9–12 h and 4 h, respectively. N-Methyl-2-pyrrolidone is hyroxylated to 5-hydroxy-N-methyl-2-pyrrolidone (5-HNMP) by the isoform 1E of cytochrome P450 (CYP1E) and oxidized to N-methylsuccinimide (MSI); MSI is then hyroxylated to 2-hydroxy-N-methylsuccinimide (2-HMSI). 2-Pyrrolidone is reported as another metabolite of N-Methyl-2-pyrrolidone [7].
In rats, it is absorbed rapidly after inhalation, oral, and dermal administration, distributed throughout the organism, and eliminated mainly by hydroxylation to polar compounds, which are excreted via urine. About 80% of the administered dose is excreted as it and its metabolites within 24 hours. A probably dose dependent yellow coloration of the urine in rodents is observed. The major metabolite is 5-hydroxy-N-methyl-2-pyrrolidone.
Toxicity
N-Methyl-2-pyrrolidone has significant cardiovascular toxicity, and the arterial pressure change induced after interarterial infusion is more than that caused by other solvents. An increase has been reported in the hemoglobin amount and leukocyte count of the peripheral blood of workers exposed to vapors of a mixture of N-dimethylacetamide and N-Methyl-2-pyrrolidone. In addition, there has been an elevation in the levels of glutamate pyruvate transaminase and glutamate oxaloacetate transaminase during the ensuing two years of surveillance. N-Methyl-2-pyrrolidone was considered responsible for the stillborn fetus, because no other possible risk factor was present that could have caused the stillbirth. In addition, studies on animals had shown that N-Methyl-2-pyrrolidone is a fetotoxic solvent, thus Gina and coworkers concluded that N-Methyl-2-pyrrolidone might be fetotoxic in humans also [8]. Exposure to 10, 25, and 50 mg/m3 N-Methyl-2-pyrrolidone does not cause eye, nose, and airway irritation. Workplace exposure to N-Methyl-2-pyrrolidone does not cause any health complaints, except in the case of persons not using personal health–protecting equipments.
References
[1]Harreus et al. Ullmann's Encyclopedia of Industrial Chemistry. 2011, Weinheim: Wiley-VCH.
[2]Smallwood. Handbook of Organic Solvents Properties. Arnold, Hodder Headline Group, London, UK, 1996.
[3]Sickley. Solubilizing excipients in oral and injectable formulations. Pharm Res, 2004, 21:201-230.
[4]Fujii et al. Preparation of griseofulvin for topical application using N-methyl-2-pyrrolidone. Biol Pharm Bull, 2001, 23:1341-1345.
[5]Sitarek et al. Tissue distribution and excretion of N-methyl-2-pyrrolidone in male and female rats. Int J Occup Med Environ Health, 2006, 19:142-148.
[6]Ursin et al. Permeability of commercial solvents through living human skin.Am Ind Hyg Assoc J, 1995, 56:651-660.
[7]Carnerup et al. Levels of N-methyl-2-pyrrolidone (NMP) and its metabolites in plasma and urine from volunteers after experimental exposure to NMP in dry and humid air. Toxicol Lett, 2006, 162:139-145.
[8]Gina et al. Hygienic studies on workers engaged in producing synthetic heatresisting enamel wire. Mixed toxicity of N-dimethylacetamide and N-methylpyrrolidone. Clinician, 1981, 45:327-338.
- Related articles
- Related Qustion
- N-Methyl-2-pyrrolidone: A Green Solvent for Coal Extraction Feb 14, 2025
N-Methyl-2-pyrrolidone (NMP) is a versatile, green industrial solvent that can be used in a variety of industries and applications such as paint and coating removal, pharmaceutical solubilizer, engineering plastic coating, etc.
- How to synthesis N-Methyl-2-pyrrolidone Mar 12, 2024
Based on the raw materials used, several processes such as succinonitrile route, condensation of γ-butyrolactone and methylamine, 4-oxidized methyl butyrate method, and 1,4-succinic acid route for manufacture of NMP were developed.
- Toxic hygroscopic contact reaction to N-methyl-2-pyrrolidone Nov 23, 2023
Exposure to organic solvents such as NMP may result in neurological toxicity, inhalation in high concentrations causing headache, giddiness, mental confusion and nausea, and ingestion abdominal pain, nausea and vomiting.
Monomethyl auristatin E is an agent with anti-mitotic activity, which is often used to inhibit cell division by blocking the polymerization of tubulin.....
Apr 11,2023Drugs2-Chlorobenzaldehyde (CAS No.: 89-98-5), also known as Amlodipine Impurity T, with molecular formula of C7H5ClO and molecular weight of 140.57, is used as medicine and dye intermediate.....
Apr 12,2023Pharmaceutical intermediatesN-Methyl-2-pyrrolidone
872-50-4You may like
N-Methyl-2-pyrrolidone manufacturers
- 1-Methyl-2-pyrrolidinone
-

- 2025-12-11
- CAS:872-50-4
- Min. Order:
- Purity: 0.99
- Supply Ability:
- N-methylpyrrolidin-2-one
-

- $1.00 / 10g
- 2025-12-11
- CAS:872-50-4
- Min. Order: 10000000g
- Purity: 99.9%
- Supply Ability: 5000
- 1-Methyl-2-pyrrolidinone
-

- $1.00 / 1kg
- 2025-12-11
- CAS:872-50-4
- Min. Order: 1kg
- Purity: 99%
- Supply Ability: 10 mt