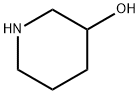
Properties, Preparation Methods, and Derivatives of 3-Hydroxypiperidine
Aug 29,2025
3-Hydroxypiperidine is a valuable intermediate used in various organic synthesis applications. This white to light yellow crystalline powder possesses a unique structure containing a piperidine ring with a hydroxyl group at the 3-position. Its diverse reactivity makes it a popular choice for researchers in the field of organic chemistry. 3-Hydroxypiperidine finds application in the synthesis of various organic compounds, including: Unsymmetrical ureas: 3-Hydroxypiperidine can be utilized as a building block for the synthesis of unsymmetrical ureas, which are important functional groups in pharmaceuticals and materials science. Piperidine nucleoside analogs: This intermediate play a role in the synthesis of piperidine nucleoside analogs, a class of compounds with potential therapeutic applications. Fluorination reactions: 3-Hydroxypiperidine can be employed in fluorination reactions, allowing for the introduction of fluorine atoms into organic molecules. Substituted pyridines: This versatile chemical serves as a starting material for the synthesis of substituted pyridines, a class of heterocyclic compounds with a wide range of applications.

Preparation method of 3-hydroxypiperidine
Piperidinol and its derivatives are important pharmaceutical intermediates. They are widely used in the synthesis of diabetes drugs, antidepressants, antitumor drugs, local anesthetics, and antihypertensive drugs, among which (S)-3-hydroxyl Due to its special structure, pyridine is widely used in pharmaceutical synthesis. At present, the most common method for preparing 3-hydroxypiperidine is direct catalytic hydrogenation of 3-hydroxypyridine. Catalytic hydrogenation commonly used catalysts include palladium carbon, rhodium carbon, ruthenium carbon and other catalysts, for the above-mentioned catalytic hydrogenation, ruthenium carbon catalyst catalytic effect is best, but the price of rhenium is relatively high (about 5 times that of ruthenium), and The pollution is relatively heavy and not suitable for industrial production. Although the price of antimony is relatively low, the catalytic performance of antimony carbon catalysts is relatively poor.[1]
In this embodiment, 3-hydroxypiperidine is prepared by catalytic hydrogenation of 3-hydroxypyridine. The specific method is as follows: 95 g of 3-hydroxypyridine (1.0 mol) and 600 mL of water were added to the autoclave, and the mixture was stirred and dissolved. Then, 5 wt % of rhodium/silica catalyst 5.0 g and cocatalyst 2.5 g of alumina were added. After three times of nitrogen substitution, hydrogen gas was introduced and the reaction was maintained at a pressure of 7 MPa and a temperature of 85° C. until complete. After the reaction, the mixture was filtered, and the filtrate was distilled off and then distilled (65-67°C, 2 mmHg) to give 99.7 g of 3-hydroxypiperidine. The yield was 98.7%, and the GC content was 99.5%.
Facile Syntheses of Enantiopure 3-Hydroxypiperidine Derivatives and 3-Hydroxypipecolic Acids
Azasugars are carbohydrate analogues in which a nitrogen atom substitutes for the oxygen atom in the ring, and they have been found in a number of plants and microorganisms. Since the first discovery of nojirimycin in 1966, azasugars have received a great deal of attention due to their significant bioactivity. Their physiological effects result from the similarity with transition-state analogues in many carbohydrate-processing enzymes such as glycosidases and glycosyltransferases. Since azasugars are viewed as possible therapeutic agents to treat a number of diseases such as diabetes, viral infections, and tumor metastasis, they have attracted synthetic chemists’ interests to develop various approaches. As a part of the project devoted to asymmetric syntheses of different azasugars and derivatives for pharmaceutical purposes, here we report the syntheses of enantiopure 3-hydroxypiperidine derivatives and 3-hydroxypipecolic acids through common intermediates via Rh-catalyzed cyclohydrocarbonylation. Facile syntheses of enantiopure trans- and cis-3-hydroxypiperidine derivatives and 3-hydroxypipecolic acids are reported, featuring Rh-catalyzed cyclohydrocarbonylation through common intermediates. A diaxial conformation in a 2,3-disubstituted N-Boc-piperidinyl structure is revealed by an X-ray crystallographic analysis.[2]
Enantioselective Construction of 3-Hydroxypiperidine Scaffolds
3-Hydroxypiperidine scaffolds were enantioselectively constructed in an atom-economical way by the title reaction. In a formal sense, the allylic CH bond was selectively cleaved and added across the ketonic carbonyl group with migration of the double bond. A piperidine scaffold is one of the structural motifs most prevalent among biologically active compounds, including natural products. Although a wide variety of methods have been developed for the construction of piperidine scaffolds, there is still a strong demand for catalytic asymmetric procedures offering straightforward access starting from simple molecules. We herein report enantioselective construction of 3-hydroxypiperidine scaffolds by the sequential action of light and rhodium upon N-allylglyoxylamides. In a formal sense, the allylic CH bond is selectively cleaved and added across the ketonic carbonyl group with migration of the double bond (carbonyl-ene type reaction), thus forming the six-membered ring with a chiral quaternary carbon center in an atom-economical way. 3-Hydroxypiperidine scaffolds were enantioselectively constructed in an atom-economical way by sequential action of light and rhodium upon N-allylglyoxylamides. In a formal sense, the allylic CH bond was selectively cleaved and enantioselectively added across the ketonic carbonyl group with migration of the double bond (carbonyl-ene-type reaction).[3]
References
[1]JIANGSU SUNLIGHT PHARMACEUTICAL CHEMICAL MAT - CN108017572, 2018, A
[2]Chiu, W.‐H., Lin, G.‐H., & Liang, C.‐W. (2010). Facile Syntheses of Enantiopure 3‐Hydroxypiperidine Derivatives and 3‐Hydroxypipecolic Acids. The Journal of Organic Chemistry, 75(5), 1748 - 1751. https://doi.org/10.1021/jo902324h
[3]Ishida, Naoki et al. “Enantioselective Construction of 3-Hydroxypiperidine Scaffolds by Sequential Action of Light and Rhodium upon N-Allylglyoxylamides.” Angewandte Chemie (International ed. in English) vol. 54,25 (2015): 7418-21. doi:10.1002/anie.201502584
- Related articles
- Related Qustion
Supplementation with pyridoxal 5'-phosphate monohydrate can synthesize neurotransmitters such as dopamine and serotonin, maintaining a healthy nervous system.....
Nov 4,2025Biochemical Engineering2-Methyl-3-furanthiol, boosts cooked meat-like aroma in fermented soy sauce, is safe via multiple assessments, and accumulates in stored orange juice .....
Aug 29,2025Chemical Materials3-Hydroxypiperidine
6859-99-0You may like
3-Hydroxypiperidine manufacturers
- 3-Hydroxypiperidine
-
- $0.00 / 1kg
- 2025-12-16
- CAS:6859-99-0
- Min. Order: 1kg
- Purity: 99%min
- Supply Ability: 100kgs
- 3-Hydroxypiperidine
-
- $5.00 / 1KG
- 2025-09-25
- CAS:6859-99-0
- Min. Order: 1KG
- Purity: 99%
- Supply Ability: g-kg-tons, free sample is available
- 3-Hydroxypiperidine
-

- $0.00 / 1kg
- 2024-06-11
- CAS:6859-99-0
- Min. Order: 1kg
- Purity: 99%
- Supply Ability: 100 tons