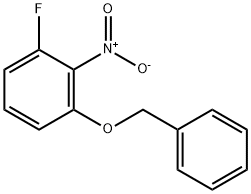
1-Benzyloxy-3-fluoro-2-nitro-benzene synthesis
- Product Name:1-Benzyloxy-3-fluoro-2-nitro-benzene
- CAS Number:920284-79-3
- Molecular formula:C13H10FNO3
- Molecular Weight:247.22
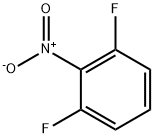
19064-24-5
277 suppliers
$6.00/1g

920284-79-3
22 suppliers
inquiry
Yield:920284-79-3 87%
Reaction Conditions:
Stage #1: benzyl alcoholwith hydrogenchloride in N,N-dimethyl-formamide at 20; for 0.333333 h;
Stage #2: 2,6-difluoro-1-nitrobenzene in N,N-dimethyl-formamide; for 1 h;
Steps:
2.1
To a suspension of sodium hydride (1.522 g, 38.06 mmol) in anhydrous DMF (150 mL) at room temperature was added benzyl alcohol (3.44 mL, 33.3 mmol) dropwise over 10 minutes, and stirring was continued for another 10 minutes. 2,6-Difluoronitrobenzene (5.046 g, 31.72 mmol) was added to the reaction mixture in one portion. The reaction mixture was stirred for an hour, and 100 mL water and 100 mL ethyl acetate were added. The layers were separated, and the aqueous layer was extracted with 75 mL ethyl acetate. The combined organic layers werre washed with 100 mL water and 100 mL brine, dried over sodium sulfate and concentrated in vacuo. The resulting residue was purified by flash chromatography (6% to 35% ethyl acetate in hexanes) to give 6.818 g (27.6 mmol, 87%) of 1-benzyloxy-3-fluoro-2-nitro-benzene as a yellow oil. MS: 248 (M+H)+. Also prepared in a similar manner using (2-hydroxy-ethyl)-methyl-carbamic acid tert-butyl ester in place of benzyl alcohol was [2-(3-Fluoro-2-nitro-phenoxy)-ethyl]-methyl-carbamic acid tert-butyl ester. MS: 215 (M-BOC+H)+.
References:
US2007/15744,2007,A1 Location in patent:Page/Page column 45

19064-24-5
277 suppliers
$6.00/1g
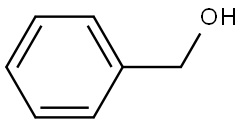
100-51-6
1432 suppliers
$5.00/100g

920284-79-3
22 suppliers
inquiry
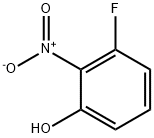
385-01-3
176 suppliers
$5.00/50mg
100-39-0
429 suppliers
$10.00/10g

920284-79-3
22 suppliers
inquiry