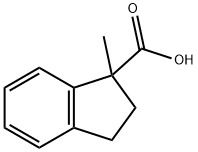
1-methyl-2,3-dihydro-1H-indene-1-carboxylic acid synthesis
- Product Name:1-methyl-2,3-dihydro-1H-indene-1-carboxylic acid
- CAS Number:33223-78-8
- Molecular formula:C11H12O2
- Molecular Weight:176.21
Yield:33223-78-8 0.45 g
Reaction Conditions:
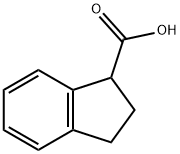
Stage #1: Indan-1-carboxylic acidwith lithium hexamethyldisilazane in tetrahydrofuran at -20 - 35; for 1 h;Inert atmosphere;
Stage #2: methyl iodide in tetrahydrofuran at 0 - 35; for 2 h;
Steps:
T-24 Example T-24: Alkylation alpha to an acid
To a solution of 2,3-dihydro-1H-indene-1-carboxylic acid (1 g, 6.17 mmol) in THF (10 mL) was added dropwise lithium bis(trimethylsilyl)amide (2.06 g, 12.34 mmol) at - 20 °C under N2. The reaction was stirred at 35 °C for 1 h. A solution of iodomethane (1.14 g, 8.02 mmol) in THF (3 mL) was added slowly at 0 °C. The reaction was stirred at 35 °C for 2 h. The mixture solution was acidified with 6 N HCl to pH = 5 and the resulting biphasic mixture was transferred to a separatory funnel. The layers were separated and the aqueous phase was extracted with EtOAc (2 x 20 mL). The combined organics were dried over anhydrous Na2SO4, filtered and concentrated in vacuo. The resulting oil was purified by flash column chromatography with an isocratic elution of EtOAc (10%) and petroleum ether (90%) to provide 1-methyl-2,3-dihydro-1H-indene-1-carboxylic acid (0.45 g, 2.55 mmol) as a brown solid.
References:
WO2018/26371,2018,A1 Location in patent:Paragraph 0217

40412-42-8
0 suppliers
inquiry

33223-78-8
10 suppliers
inquiry