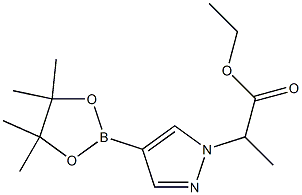
Ethyl 2-(4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-1H-pyrazol-1-yl)propanoate synthesis
- Product Name:Ethyl 2-(4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-1H-pyrazol-1-yl)propanoate
- CAS Number:1220968-24-0
- Molecular formula:C14H23BN2O4
- Molecular Weight:294.15
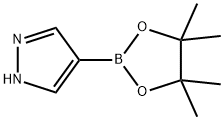
269410-08-4
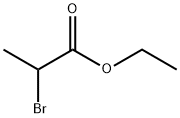
535-11-5

1220968-24-0
The reaction was carried out in the presence of cesium carbonate (1.5 g, 4.6 mmol) in acetonitrile (8 mL) at 90 °C with stirring for 4 h. The reaction was carried out in the presence of cesium carbonate (1.5 g, 4.6 mmol) at 90 °C for 4 h. The reaction was completed by cooling to room temperature and quenching the reaction with aqueous Na2CO3. After completion of the reaction, the reaction was cooled to room temperature, quenched with aqueous Na2CO3, extracted with ethyl acetate (3 × 20 mL), and the organic phases were combined and washed with brine. The organic layer was dried over anhydrous MgSO4, filtered and concentrated under reduced pressure. The crude product was purified by fast chromatography on a silica gel column eluting with gradient of ethyl acetate/hexane (0-50%) to afford the target compound ethyl 2-(4-(4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-1H-pyrazol-1-yl)propionate (0.42 g, 61% yield).LCMS (M+H)+: m/z=295.1.

269410-08-4
397 suppliers
$5.00/1g
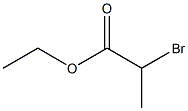
41978-69-2
2 suppliers
inquiry

1220968-24-0
35 suppliers
$65.00/100mg
Yield:1220968-24-0 61%
Reaction Conditions:
with caesium carbonate in acetonitrile at 90; for 4 h;
Steps:
54.1
A mixture of 4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-1H-pyrazole (0.40 g, 2.1 mmol, Aldrich, Cat. 525057), ethyl 2-bromopropanoate (290 μL, 2.3 mmol), and cesium carbonate (1.5 g, 4.6 mmol) in acetonitrile (8 mL) was stirred at 90° C. for 4 h. After cooling, the reaction mixture was worked up with aqueous Na2CO3, extracted with ethyl acetate (3×20 mL), and washed with brine. The combined organic layers were dried over MgSO4, filtered and concentrated under reduced pressure. The residue was purified by flash chromatography on a silica gel column with ethyl acetate in hexanes (0-50%) to afford the desired product (0.42 g, 61%). LCMS (M+H)+: m/z=295.1.
References:
US2012/165305,2012,A1 Location in patent:Page/Page column 43

41978-69-2
2 suppliers
inquiry

1220968-24-0
35 suppliers
$65.00/100mg