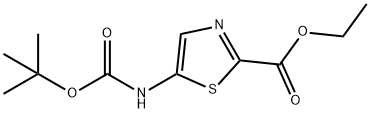
5-tert-Butoxycarbonylamino-thiazole-2-carboxylic acid ethyl ester synthesis
- Product Name:5-tert-Butoxycarbonylamino-thiazole-2-carboxylic acid ethyl ester
- CAS Number:1246549-82-5
- Molecular formula:C11H16N2O4S
- Molecular Weight:272.32
100114-63-4
12 suppliers
inquiry
24424-99-5
869 suppliers
$13.50/25G

1246549-82-5
9 suppliers
inquiry
Yield:1246549-82-5 88.2%
Reaction Conditions:
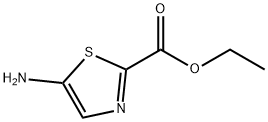
Stage #1: 5-amino-thiazole-2-carboxylic acid ethyl esterwith dmap in tetrahydrofuran at 20; for 0.25 h;
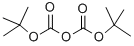
Stage #2: di-tert-butyl dicarbonate in tetrahydrofuran at 20; for 6 h;
Steps:
1 Preparation of intermediate compound A:
First, in a 100mL three-necked flask, add anhydrous tetrahydrofuran (20mL),Ethyl 5-aminothiazole-2-carboxylate (10mmol, 1.72g), 4-dimethylaminopyridine (2mmol, 0.244g),Then mix and stir at room temperature for 15 minutes, and then add di-tert-butyl dicarbonate (12mmol, 2.62g) in anhydrous tetrahydrofuran solution (10mL) dropwise,After the dropwise addition was completed, the reaction was carried out at room temperature for 6 hours, and TCL monitored (developing solvent: EA/PE=1:3) until the reaction was completed.After the completion of the reaction, spin off part of the solvent, filter, and wash the filter cake with tetrahydrofuran (3mL×2).After finally drying, a yellow solid (Compound A) is obtained.
References:
CN113024536,2021,A Location in patent:Paragraph 0133-0138

1357247-55-2
9 suppliers
inquiry

1246549-82-5
9 suppliers
inquiry