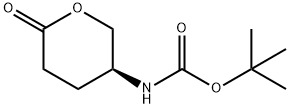
tert-butyl (S)-(6-oxotetrahydro-2H-pyran-3-yl)carbamate synthesis
- Product Name:tert-butyl (S)-(6-oxotetrahydro-2H-pyran-3-yl)carbamate
- CAS Number:125982-23-2
- Molecular formula:C10H17NO4
- Molecular Weight:215.25
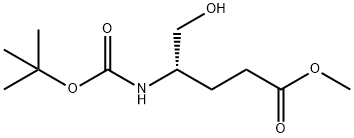
126587-35-7

125982-23-2
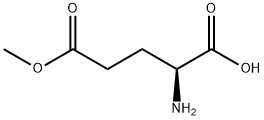
Methyl (S)-4-((tert-butoxycarbonyl)amino)-5-hydroxypentanoate (25 g, 0.1 mol, 1.0 eq.) was used as a raw material, which was dissolved in a mixed solvent of toluene (300 mL) and acetic acid (150 mL), and the reaction was heated and refluxed for 5 hours. Upon completion of the reaction, the reaction solution was cooled to room temperature and subsequently concentrated under reduced pressure to remove the solvent. The concentrated residue was placed in an ice bath and the pH was adjusted to 7-8 by slowly adding saturated sodium bicarbonate solution. the aqueous phase was extracted three times with ethyl acetate, and the organic phases were combined, washed with saturated brine and dried over anhydrous sodium sulfate. After filtration, the filtrate was concentrated to give the crude product. The crude product was purified by solvent recrystallization with mixed solvents of ethyl acetate and petroleum ether to afford the target product tert-butyl (S)-(6-oxotetrahydro-2H-pyran-3-yl)carbamate (8.0 g, 37.2% yield) as a white powder. The product was analyzed by GC-MS and the molecular ion peak was 215.

126587-35-7
38 suppliers
$45.00/10mg

125982-23-2
43 suppliers
inquiry
Yield:125982-23-2 37.2%
Reaction Conditions:
with acetic acid in toluene; for 5 h;Reflux;
Steps:
4.3 Step 3
A solution of compound of A4-3 (25g, O. lmol, l .Oeq) in toluene (300mL) and acetic acid (150mL) was heated to reflux for 5hrs and then cooled, concentrated under vacuum. The residual was added saturated sodium bicarbonate solution to pH 7-8 in ice-bath. Then the mixture was extracted three times with ethyl acetate, and the combined extracts was washed with brine, dried over anhydrous sodium sulfate, concentrated and recrystallized by ethyl acetate and PE to give compound A4-4 as a white powder (8.0g, yield 37.2%). GC-MS: 215
References:
WO2018/67422,2018,A1 Location in patent:Page/Page column 47; 48
24424-99-5
853 suppliers
$13.50/25G

125982-23-2
43 suppliers
inquiry
45214-91-3
200 suppliers
$10.00/5g

125982-23-2
43 suppliers
inquiry
1499-55-4
307 suppliers
$6.00/5g

125982-23-2
43 suppliers
inquiry

39538-20-0
4 suppliers
inquiry

125982-23-2
43 suppliers
inquiry