
(S)-4-(4,6-Dichloropyrimidin-2-yl)-3-methylmorpholine synthesis
- Product Name:(S)-4-(4,6-Dichloropyrimidin-2-yl)-3-methylmorpholine
- CAS Number:1333108-98-7
- Molecular formula:C9H11Cl2N3O
- Molecular Weight:248.11
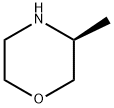
350595-57-2
169 suppliers
$10.00/1g
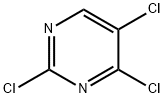
5750-76-5
360 suppliers
$5.00/1g

1012317-33-7
1 suppliers
inquiry

1333108-98-7
5 suppliers
inquiry
Yield:1012317-33-7 69% ,1333108-98-7 22%
Reaction Conditions:
with N-ethyl-N,N-diisopropylamine in dichloromethane at 0 - 20; for 3 h;
Steps:
1A; 1B
To a solution of trichloropyrimidine (5.04g, 27.3mol) in DCM (50mL) cooled to 0°C was added DIPEA (4.79mL, 27.3mol), followed by 3-(S)-methylmorpholine (3.1 lg, 30.5mol) dropwise. The reaction mixture was allowed to warm to RT and stirred for 3h wherupon it was diluted with DCM, washed with water and brine, dried over magnesium sulfate, filtered and the solvent removed in vacuo. The residue was purified by flash chromatography (5-10% EtOAc in petroleum ether (40-60)) to yield the two isomers. Intermediate 1A as a white solid 4.66g, 69%. LCMS (method B), (M+H+) 248, Rt = 2.48min.Intermediate IB as white crystals 1.5g, 22%. LCMS (method B), (M+H+) 248, Rt = 3.00min.
References:
WO2011/107585,2011,A1 Location in patent:Page/Page column 40-41

350595-57-2
169 suppliers
$10.00/1g
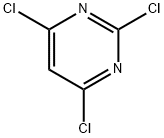
3764-01-0
400 suppliers
$6.00/5g

1333108-98-7
5 suppliers
inquiry

350595-57-2
169 suppliers
$10.00/1g

1333108-98-7
5 suppliers
inquiry