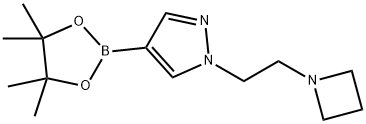
1-[2-(azetidin-1-yl)ethyl]-4-(tetramethyl-1,3,2-dioxaborolan-2-yl)-1H-pyrazole synthesis
- Product Name:1-[2-(azetidin-1-yl)ethyl]-4-(tetramethyl-1,3,2-dioxaborolan-2-yl)-1H-pyrazole
- CAS Number:1422126-12-2
- Molecular formula:C14H24BN3O2
- Molecular Weight:277.17
503-29-7
182 suppliers
$35.00/1g
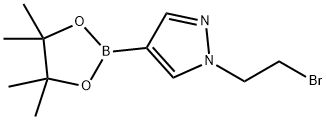
1111269-34-1
12 suppliers
inquiry
![1-[2-(azetidin-1-yl)ethyl]-4-(tetramethyl-1,3,2-dioxaborolan-2-yl)-1H-pyrazole](/CAS/20180629/GIF/1422126-12-2.gif)
1422126-12-2
32 suppliers
$165.00/100MG
Yield:1422126-12-2 49.6 mg
Reaction Conditions:
with potassium carbonate in acetonitrile at 20 - 60; for 18 h;
Steps:
24 (Synthesis of Compound 0024-1)
- Synthesis of Compound 0024 -
(Synthesis of Compound 0024-1)
[Chem. 52]
Azetidine (37.9 mg) was added to a suspension of 1-(2-bromoethyl)-1H-pyrazole-4-boronic acid pinacol ester (which was synthesized according to ) (99.8 mg) and potassium carbonate (91.8 mg) in acetonitrile (0.3 mL), and the mixture was stirred at room temperature for 15 hours and at 60°C for 3 hours. The reaction mixture was returned to room temperature, ethyl acetate and saturated aqueous sodium bicarbonate were added thereto, and the resulting mixture was extracted with ethyl acetate. The organic layer was washed with water and brine, and then dried over sodium sulfate. The solvent was evaporated under reduced pressure and the obtained residue was purified by silica gel column chromatography (hexane-ethyl acetate) to obtain a compound 0024-1 (49.6 mg) as a colorless oily substance. 1H-NMR (CDCl3) δ: 7.78 (1H, s), 7.72 (1H, s), 4.09 (2H, t), 3.15 (4H, t), 2.86 (2H, t), 2.13-1.92 (2H, m), 1.31 (12H, s). MS m/z (M+H): 278.
References:
EP2727920,2014,A1 Location in patent:Paragraph 0333-03334