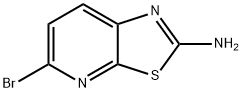
2-AMINO-5-BROMOTHIAZOLO[5,4-B]PYRIDINE synthesis
- Product Name:2-AMINO-5-BROMOTHIAZOLO[5,4-B]PYRIDINE
- CAS Number:934266-82-7
- Molecular formula:C6H4BrN3S
- Molecular Weight:230.09
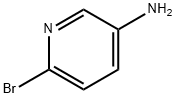
13534-97-9
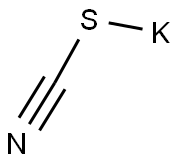
333-20-0
![2-AMINO-5-BROMOTHIAZOLO[5,4-B]PYRIDINE](/CAS/GIF/934266-82-7.gif)
934266-82-7
Step A: 6-Bromopyridin-3-amine (1A, 20 g, 1.0 eq.) was added batchwise to a solution of potassium thiocyanate (5.0 eq.) in acetic acid (0.4 M) at -5 °C. The reaction mixture was cooled to -15 °C and a solution of bromine (Br2, 1.3 eq.) in acetic acid (9.4 M) was slowly added dropwise through a constant pressure dropping funnel. After the dropwise addition, the reaction system was slowly warmed to room temperature and stirred continuously for 12 hours. Upon completion of the reaction, the precipitate was collected by filtration. To the filtrate, 100 mL of ethyl acetate (EtOAc) and 200 mL of water (H2O) were added, followed by concentration under reduced pressure to remove 200 mL of solvent by rotary evaporator. The residue was placed in an ice bath with stirring for 10 min, precipitated as a solid, filtered, and washed twice with a cold 10% methanol (MeOH) solution in ether (Et2O). The filtrates were combined and concentrated, and the product was crystallized in an ice bath and washed again twice with cold 10% ether solution of methanol to give a second batch of product. After vacuum drying, the brown solid product 1B was obtained in 77% yield. The product could be used for subsequent reactions without further purification. The product was characterized as follows: 1H NMR (400 MHz, DMSO-d6) δ ppm 4.94 (br.s, 1H), 7.44 (d, J = 8.34 Hz, 1H), 7.57 (d, J = 8.34 Hz, 1H), 8.04 (br.s., 1H); ESI-MS: m/z 230.0 (M + H)+.
![N-(5-BroMothiazolo[5,4-b]pyridin-2-yl)acetaMide](/CAS/GIF/1112982-76-9.gif)
1112982-76-9
27 suppliers
$45.00/50mg
![2-AMINO-5-BROMOTHIAZOLO[5,4-B]PYRIDINE](/CAS/GIF/934266-82-7.gif)
934266-82-7
65 suppliers
$23.00/250mg
Yield:934266-82-7 93%
Reaction Conditions:
Stage #1: N-(5-bromothiazolo[5,4-b]pyridin-2-yl)acetamidewith hydrogenchloride;water for 1 h;Reflux;
Stage #2: in water at 20; pH=10;
Steps:
4; 4.4-ii
As shown in step 4-ii of Scheme 21, Compound 1005 (5.44 g, 20 mmol) was suspended in 6 N HCl (100 mL). The reaction mixture was heated at reflux for 1 h, at which time all of the material went into solution. The mixture was cooled to RT and the reaction was made basic to a pH of 10, at which time the product precipitated out. The solid was collected on a fritted funnel and dried to afford 5-bromothiazolo[5,4-δ]pyridin-2-amine (Compound 1006, 4.35 g, 93% yield): ESMS (M+H) 230, 232; 1H NMR (DMSO-d6) δ 7.9 (br, 2H), 7.6 (d, IH), 7.4 (d, IH).
References:
WO2009/129211,2009,A1 Location in patent:Page/Page column 35; 37

13534-97-9
437 suppliers
$6.00/1g

333-20-0
463 suppliers
$13.50/100G
![2-AMINO-5-BROMOTHIAZOLO[5,4-B]PYRIDINE](/CAS/GIF/934266-82-7.gif)
934266-82-7
65 suppliers
$23.00/250mg

13534-97-9
437 suppliers
$6.00/1g
![2-AMINO-5-BROMOTHIAZOLO[5,4-B]PYRIDINE](/CAS/GIF/934266-82-7.gif)
934266-82-7
65 suppliers
$23.00/250mg

169833-70-9
130 suppliers
$9.00/250mg
![2-AMINO-5-BROMOTHIAZOLO[5,4-B]PYRIDINE](/CAS/GIF/934266-82-7.gif)
934266-82-7
65 suppliers
$23.00/250mg
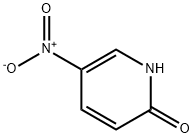
5418-51-9
449 suppliers
$12.72/1gm:
![2-AMINO-5-BROMOTHIAZOLO[5,4-B]PYRIDINE](/CAS/GIF/934266-82-7.gif)
934266-82-7
65 suppliers
$23.00/250mg