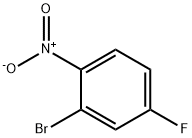
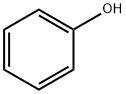
2-BROMO-1-NITRO-4-PHENOXYBENZENE synthesis
- Product Name:2-BROMO-1-NITRO-4-PHENOXYBENZENE
- CAS Number:1258545-24-2
- Molecular formula:C12H8BrNO3
- Molecular Weight:294.1
Yield:1258545-24-2 94%
Reaction Conditions:
Stage #1: phenolwith caesium carbonate in acetonitrile at 20; for 0.166667 h;
Stage #2: 2-bromo-4-fluoronitrobenzene in acetonitrile at 50; for 65 h;
Steps:
71
Example 71; Synthesis of (3S)-3-amino-1-hydroxy-6-phenoxy-3,4-dihydroquinolin-2(1H)-one, hydrochloride salt (93); 2-Bromo-1-nitro-4-phenoxybenzene (90); Phenol (11.1 g, 118 mmol) was added to a suspension of Cs2CO3 (46.2 g, 142 mmol) in MeCN (295 mL). The resulting solution was stirred at RT for 10 min, then 2-bromo-4-fluoronitrobenzene (26.0 g, 118 mmol) was added, and the reaction mixture was heated to 50° C. for 65 h. The reaction mixture was cooled to RT and filtered to remove Cs2CO3. The filtrate was concentrated in vacuo, and the resulting residue was dissolved in EtOAc (150 mL) and washed with aqueous sodium hydroxide solution (1 N, 250 mL), water (2×250 mL), and saturated aqueous sodium chloride solution (250 mL). The separated organic phase was dried over Na2SO4, filtered, and concentrated in vacuo. Purification by silica gel chromatography (Eluant: heptane) provided the title compound as a pale yellow oil (32.7 g, 94%). 1H NMR (400 MHz, CDCl3) δ 6.97 (dd, J=9.1, 2.6 Hz, 1H), 7.08-7.12 (m, 2H), 7.26-7.31 (m, 2H), 7.43-7.49 (m, 2H), 7.95 (d, J=9.1 Hz, 1H).
References:
US2010/324043,2010,A1 Location in patent:Page/Page column 44