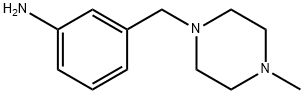
3-(4-METHYL-PIPERAZIN-1-YLMETHYL)-ANILINE synthesis
- Product Name:3-(4-METHYL-PIPERAZIN-1-YLMETHYL)-ANILINE
- CAS Number:198281-55-9
- Molecular formula:C12H19N3
- Molecular Weight:205.3
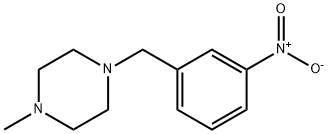
198281-54-8
37 suppliers
inquiry

198281-55-9
52 suppliers
$35.35/250mgs:
Yield:198281-55-9 95%
Reaction Conditions:
with hydrogen
Steps:
23.A
A. 3-(4-Methyl-piperazin-1-ylmethyl)-phenylamine m-Nitrobenzyl bromide (500 mg, 2.31 mmol) was added to a mixture of 1-methylpiperazine (277 mg, 2.77 mmol) and potassium carbonate (414 g, 3.0 mmol) in 5 mL of DMF. The mixture was stirred at 90° C. for 16 hours. After cooling to room temperature, the mixture was extracted into ethyl acetate (EtOAc), washed with water, brine and then dried with sodium sulfate (Na2SO4). Removal of the solvent and chromatography on silica, eluding with EtOAc/CH3OH/NH4OH (10:1:0.1, v/v), gave 510 mg of 1-methyl-4-(3-nitro-benzyl)-piperazine, which was converted to the title compound under normal hydrogenation conditions. The titled compound was obtained as a yellow solid (450 mg, 95%). 1H NMR (400 MHz, CDCl3) δ (ppm): 7.02 (t, J=7.6 Hz, 1H), 6.62 (s, 1H), 6.60 (d, J=7.6 Hz, 1H), 6.54 (d, J=7.6 Hz, 1H), 3.43 (s, 2H), 2.76 (br, 4H), 2.65 (br, 4H), 2.82 (m, 4H), 2.46 (s, 3H).
References:
US2007/60577,2007,A1 Location in patent:Page/Page column 42
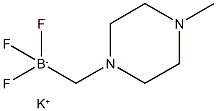
1015484-22-6
57 suppliers
$33.00/1g
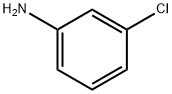
108-42-9
476 suppliers
$10.00/5g

198281-55-9
52 suppliers
$35.35/250mgs:
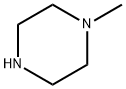
109-01-3
672 suppliers
$5.00/5G

198281-55-9
52 suppliers
$35.35/250mgs:

109-01-3
672 suppliers
$5.00/5G
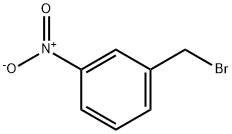
3958-57-4
252 suppliers
$14.00/10g

198281-55-9
52 suppliers
$35.35/250mgs:

3958-57-4
252 suppliers
$14.00/10g

198281-55-9
52 suppliers
$35.35/250mgs: