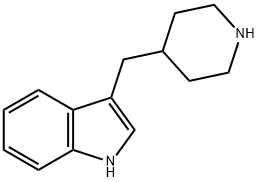
3-(4-Piperidylmethyl)-1H-indole synthesis
- Product Name:3-(4-Piperidylmethyl)-1H-indole
- CAS Number:3515-49-9
- Molecular formula:C14H18N2
- Molecular Weight:214.31
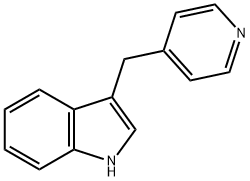
5275-07-0

3515-49-9
The general procedure for the synthesis of 3-(piperidin-4-ylmethyl)-1H-indole from 3-(4-pyridinylmethyl)-1H-indole was as follows: 3-(4-pyridinylmethyl)-1H-indole (5.00 g, 24.0 mmol) and acetic acid (25 mL) were added to a 250 mL stainless steel autoclave, followed by platinum(IV) oxide (1.00 g, 4.40 mmol, Johnson Matthey). The autoclave was sealed, charged with hydrogen to about 30 psi at ambient temperature, and the reaction was stirred for about 2 hours. Upon completion of the reaction, the reaction mixture was filtered to remove the catalyst and the filtrate was concentrated under reduced pressure. The concentrated residue is dissolved in ethyl acetate and subsequently washed with saturated aqueous sodium carbonate. At this point the organic layer may appear cloudy and a small amount of methanol is added until the solution is clarified. Next, the organic layer was washed sequentially with 2N aqueous sodium hydroxide and saturated brine, dried over anhydrous magnesium sulfate and filtered, and the filtrate was concentrated under reduced pressure. The resulting solid was ground with ether and dried to give the target product 3-(piperidin-4-ylmethyl)-1H-indole (4.35 g, 73% yield). The product was analyzed by LC/MS (see Table 1 for conditions, method a) with a retention time (Rt) of 1.51 min; the mass spectrum (MS) showed m/z: 215.12 ([M+H]+).

5275-07-0
12 suppliers
$599.00/1g

3515-49-9
20 suppliers
inquiry
Yield:3515-49-9 73%
Reaction Conditions:
Stage #1: 3-<(4-pyridyl)methyl>-1H-indolewith hydrogen;acetic acid;platinum(IV) oxide at 20; under 1551.49 Torr; for 2 h;
Stage #2: with sodium carbonate in water;ethyl acetate;
Steps:
8
3-(Pyridin-4-ylmethyl)-1H-indole (5.00 g, 24.0 mmol) and acetic acid (25 mL) were added to platinum(IV) oxide (1.00 g, 4.40 mmol, Johnson Matthey) in a 250 mL stainless steel pressure bottle and stirred for about 2 hr under about 30 psi hydrogen at ambient temperature. The mixture was filtered and the filtrate was concentrated under reduced pressure. The residue was dissolved with EtOAc and then washed with saturated aqueous Na2CO3. The organic layer became turbid. A small amount of MeOH was added to form a clear solution. The organic layer was washed with 2 N aqueous NaOH and brine, dried over MgSO4, filtered, and concentrated under reduced pressure. The resulting solid was triturated with Et2O and then dried to give 3-(piperidin-4-ylmethyl)-1H-indole (4.35 g, 73% yield): LC/MS (Table 1, Method a) Rt=1.51 min; MS m/z: 215.12 (M+H)+.
References:
US2011/152243,2011,A1 Location in patent:Page/Page column 41-42

63845-36-3
0 suppliers
inquiry

3515-49-9
20 suppliers
inquiry

51626-60-9
1 suppliers
inquiry

3515-49-9
20 suppliers
inquiry
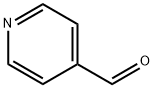
872-85-5
495 suppliers
$10.00/1g

3515-49-9
20 suppliers
inquiry
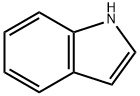
120-72-9
659 suppliers
$6.00/25g

3515-49-9
20 suppliers
inquiry