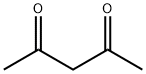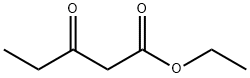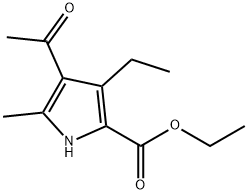
1H-Pyrrole-2-carboxylic acid, 4-acetyl-3-ethyl-5-Methyl-, ethyl ester synthesis
- Product Name:1H-Pyrrole-2-carboxylic acid, 4-acetyl-3-ethyl-5-Methyl-, ethyl ester
- CAS Number:37013-86-8
- Molecular formula:C12H17NO3
- Molecular Weight:223.27
Yield:37013-86-8 65%
Reaction Conditions:
Stage #1: ethyl propanoylacetatewith acetic acid;sodium nitrite in water at 0 - 20; for 3 h;
Stage #2: acetylacetonewith zinc in water at 0 - 85; for 4 h;Knorr Pyrrole Synthesis;
Steps:
General procedure A for the synthesis of compounds 6a - 6g
General procedure: To a stirred ice-cooled solution of compound 1a - 1e (20mmol) in HOAc (40mL), the sodium nitrite (30mmol) in H2O (5mL) was added dropwise. The reaction mixture was stirred at r.t. for about 3h, then Zn (40mmol) and pentane-2,4-dione (20mmol) was added at 0°C. And the reaction mixture was heated to 85°C for about 4h. Upon completion, the reaction mixture was poured into water and was extracted with ethyl acetate (100mL×3). The combined organic layers were washed with brine, dried over Na2SO4, and concentrated under vacuum. The residue was chromatographed, eluting with a gradient of 2-10% EtOAc/petroleum ether to afford compound 2a - 2e (yield, 58-89%) as a clear oil. (0047) To a suspension of compound 2a - 2e (10mmol), KOH (50mmol) in ethylene glycol (20mL). The resulting mixture was stirred at 140°C for about 3h. After completion of reaction, the mixture was poured into water and was extracted with ethyl acetate (100mL×3). The combined organic layers were washed with brine, dried over Na2SO4, and concentrated under vacuum to give 3a - 3e (yield, 72-91%) without further purification. (0048) To a solution of 3a (2.0g, 14.6mmol) in THF (20mL) at - 78°C was added NBS (2.6g, 14.6mmol) in four batches. The reaction mixture was stirred at - 78°C for 1h. Then the reaction was partitioned between water and ethyl acetate, the aqueous layer was extracted with additional ethyl acetate twice. The combined organic layers was washed with brine, dried over Na2SO4, filtered, and concentrated. The residue was chromatographed, eluting with a gradient of 10-20% EtOAc/petroleum ether to afford compound 4 (1.8g, yield, 57%) as a yellow solid. (0049) A mixture of 3a - 3e (10mmol) in anhydrous DMF (20mL) was stirred at 0°C, the solution was added dropwise POCl3 (20mmol). The mixture was stirred at ambient temperature for about 3h. Then the mixture was added dropwise 5M KOAc (50mmol) solution at 0°C, and the solution was stirred at 105°C under nitrogen for about 3h. After cooling, the mixture was poured into water and was extracted with ethyl acetate (100mL×3). The combined organic layers were washed with brine, dried over Na2SO4, filtered, and concentrated. The resulting crude product was purified by silica gel column chromatography (10-30% EtOAc/petroleum ether) to yield 5a - 5e (yield, 51-74%) as a brown solid. (0050) A flask was charged with compound 4 (0.22g, 1mmol), corresponding boric ester (1.2mmol), PdCl2(dppf) (0.1mmol), K2CO3 (5mmol) and sparged with nitrogen. Degassed dioxane (7mL) and water (2mL) was added and the mixture was heated at 100°C for 4h. The reaction mixture was partitioned between water and ethyl acetate, the aqueous layer was extracted with additional ethyl acetate twice. The combined organic layers were washed with brine, dried over Na2SO4, filtered, and concentrated. The crude product was purified by silica gel column chromatography (30-80% EtOAc/petroleum ether) to yield 6a - 6e (yield, 49-68%) as a white solid. (0051) A mixture of 5a (0.2g, 1.2mmol), Na2S2O5 (0.023g, 0.12mmol), benzene-1,2-diamine (0.13g, 1.2mmol) or 2-aminobenzenethiol (0.15g, 1.2mmol) in DMF (10mL) was degassed under a stream of nitrogen. The resulting brown solution was heated at 140°C overnight. The cooled reaction was purified by silica gel column chromatography (gradient elution, 1-2% MeOH/dichloromethane) to afford 6f - 6g (yield, 59, 62%) as a white solid.
References:
Kong, Bo;Zhu, Zhaohong;Li, Hongmei;Hong, Qianqian;Wang, Cong;Ma, Yu;Zheng, Wan;Jiang, Fei;Zhang, Zhimin;Ran, Ting;Bian, Yuanyuan;Yang, Na;Lu, Tao;Zhu, Jiapeng;Tang, Weifang;Chen, Yadong [European Journal of Medicinal Chemistry,2022,vol. 227,art. no. 113953]