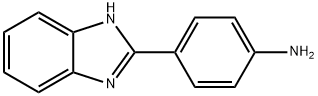
4-(1H-BENZOIMIDAZOL-2-YL)-PHENYLAMINE synthesis
- Product Name:4-(1H-BENZOIMIDAZOL-2-YL)-PHENYLAMINE
- CAS Number:2963-77-1
- Molecular formula:C13H11N3
- Molecular Weight:209.25
150-13-0
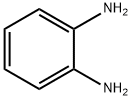
95-54-5

2963-77-1
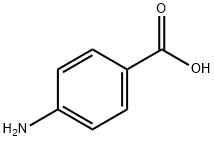
The reaction was carried out with 4-aminobenzoic acid (5 g, 5.78 mmol) and o-phenylenediamine (3.9 g, 3.68 mmol) in polyphosphoric acid (12.5 g) with stirring at 200 °C for 5 hours. Upon completion of the reaction, the mixture was cooled to room temperature and slowly poured into crushed ice. Subsequently, the precipitate was stirred in cold water. The pH was adjusted with ammonium hydroxide solution to 7. The solid product was collected by filtration and washed several times with methanol. Finally, the target compound 4-(1H-benzimidazol-2-yl)aniline (5) was purified as a white solid by silica gel column chromatography using ethyl acetate: hexane (4:1) as eluent. Yield: 82%; Melting point: 207-209 °C; 1H NMR (400 MHz, DMSO-d6): δ= 7.96 (d, 2H, J = 8.72 Hz, ArH), 7.55-7.51 (m, 2H, ArH), 7.16-7.14 (m, 2H, ArH), 6.75 (d, 2H, J = 8.24 Hz, ArH) , 4.45 (bs, 2H, NH2); 13C NMR (100 MHz, DMSO-d6): δ= 152.2,148.4,127.5,120.9,118.6,113.8 (ArC); MS (ESI), m/z: 210.2 (M++1).
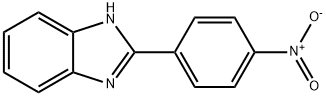
729-13-5
29 suppliers
$160.00/0.500g

2963-77-1
91 suppliers
$45.00/10mg
Yield: 95%
Reaction Conditions:
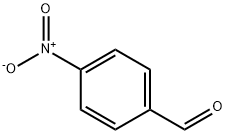
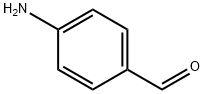
with hydrogenchloride;iron in ethanol;waterReflux;
Steps:
General procedure: Iron powder (3-4 mmol) and 6 N HCl solution (1 ml) were added to the solution of the nitro derivatives (1 mmol) in EtOH (5 ml) and the mixture was heated to reflux under nitrogen for 3-5 h. After cooling to room temperature, the suspension was diluted with EtOAc (20 ml); a solution of 6 M NaOH (20 ml) was added until basic pH and the layers were separated. The aqueous layer was extracted with AcOEt(2 X 20 ml) and the combined organic layers were washed with brine,dried over anhydrous Na2SO4, filtered and evaporated under vacuum.The crude product was purified by chromatography on silica gel(eluent: EtOAc/Hexane 6:4 v/v or CHCl3/MeOH 9.5:0.5 v/v to 9:1 v/v)or triturated with diethyl ether.
References:
Agamennone, Mariangela;Caradonna, Alessia;Di Pizio, Antonella;Laghezza, Antonio;Loiodice, Fulvio;Luisi, Grazia;Piemontese, Luca;Tortorella, Paolo [Bioorganic and medicinal chemistry,2019]
619-73-8
576 suppliers
$5.00/10g

95-54-5
541 suppliers
$9.00/1g

2963-77-1
91 suppliers
$45.00/10mg

729-13-5
29 suppliers
$160.00/0.500g