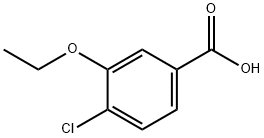
4-CHLORO-3-ETHOXYBENZOIC ACID synthesis
- Product Name:4-CHLORO-3-ETHOXYBENZOIC ACID
- CAS Number:97209-05-7
- Molecular formula:C9H9ClO3
- Molecular Weight:200.62
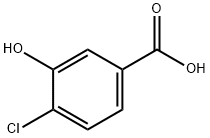
34113-69-4
144 suppliers
$6.00/250mg
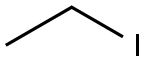
75-03-6
369 suppliers
$11.00/5g

97209-05-7
16 suppliers
$65.00/250mg
Yield:97209-05-7 91%
Reaction Conditions:
with potassium carbonate in N,N-dimethyl-formamide at 20; for 6 h;
Steps:
1.1
To a solution of 4-chloro-3-hydroxy-benzoic acid (3.0 g, 17.4 mmol, 1.0 equiv) in DMF (15 mL) was added K2CO3 (4.81 g, 34.8 mmol, 2.0 equiv) and ethyl iodide (4.03 mL, 5.97 g, 38.2 mmol, 2.2 equiv). The reaction mixture was stirred for 6 h at rt, diluted with water (20 mL) and extracted, with ethyl acetate (3×50 mL). The organic phase was dried over Na2SO4 and concentrated to afford 3.6 g (91%) of 4-chloro-3-ethoxy-benzoic acid ethyl ester. The crude ester was then dissolved in THF (20 mL) and cooled to -78° C. under Ar. A solution of diisobutylaluminum hydride (95 ml, 95.0 mmol, 6.0 equiv; 1.0 M solution in THF) was slowly added over a time period of 15 min, the cooling bath removed after completion of addition and the reaction allowed to reach 0° C. After stirring for 1 h, the reaction was cooled to -78° C. and the excess of hydride quenched by cautious addition of a solution of 1 M HCl (10 mL). The mixture was warmed up to rt, the organic phase separated and the aqueous layer extracted with ethyl acetate (3×100 mL). The combined organic phases were dried over Na2SO4 and concentrated by evaporation under reduced pressure providing 2.94 g (100%) of 4-chloro-3-ethoxy-benzyl alcohol. The crude alcohol (2.94 g, 15.75 mmol, 1.0 equiv) was dissolved in dichloromethane (15 mL) and activated MnO2 (5.48 g, 63.0 mmol, 4.0 equiv) was added. The reaction mixture was stirred for 16 h, after which time the reaction was filtered through Hyflo Super Cel and concentrated. The residue was purified by flash column chromatography on silica eluting with heptane/ethyl acetate (4:1) to yield 1.51 g (52%) of the title compound. 1H NMR (300 MHz, CDCl3): δ 1.51 (t, J=7.1 Hz, 3H), 4.19 (q, J=7.1 Hz, 2H), 7.37-7.42 (m, 2H), 7.55 (d, J=9.0 Hz, 1H), 9.94 (s, 1H).
References:
US2008/249101,2008,A1 Location in patent:Page/Page column 24

34113-69-4
144 suppliers
$6.00/250mg

97209-05-7
16 suppliers
$65.00/250mg