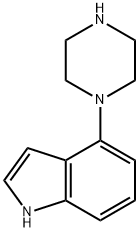
4-(Piperazino)indole synthesis
- Product Name:4-(Piperazino)indole
- CAS Number:84807-09-0
- Molecular formula:C12H15N3
- Molecular Weight:201.27
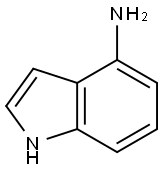
5192-23-4
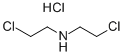
821-48-7

84807-09-0
To the reaction solution, 4-aminoindole (2 g, 15.2 mmol), bis(2-chloroethyl)amine hydrochloride (3.2 g, 18.2 mmol) and potassium carbonate (4.2 g, 30.4 mmol) were added sequentially to isopropanol (30 mL). The reaction mixture was stirred at 90 °C for 48 hours. Upon completion of the reaction, the mixture was diluted by adding dichloromethane (50 mL) and methanol (50 mL) to the mixture. The reaction mixture was filtered to remove insoluble matter and the filtrate was concentrated under reduced pressure and dried by rotary evaporator. Finally, the residue was purified using silica gel column chromatography (eluent ratio of dichloromethane/methanol (v/v) = 10/1) to afford the target product 4-(1-piperazinyl)-1H-indole as a brown solid (2.78 g, 90.8% yield).

5192-23-4
294 suppliers
$10.00/250mg

821-48-7
570 suppliers
$6.00/25g

84807-09-0
81 suppliers
$97.00/250mg
Yield:84807-09-0 90.8%
Reaction Conditions:
with potassium carbonate in isopropyl alcohol at 90; for 48 h;
Steps:
1.1 Step 1) Synthesis of 4-(piperazin-1-yl)-1H-indole
4-aminoindole (2g, 15.2mmol), bis(2-chloroethyl)amine hydrochloride (3.2g, 18.2mmol) and potassium carbonate (4.2g, 30.4mmol) was added to isopropanol (30 mL) after the reaction solution was reacted at 90 °C for 48 hours, and thereto was added dichloromethane (50mL) and methanol (50mL). The reaction mixture was filtered and the filtrate under reduced pressure to spin dry, the residue was purified by silica gel column chromatography (dichloromethane/methanol (v/v)= 10/1) by to give the title compound as a brown solid (2.78g, 90.8%).
References:
CN105367472,2016,A Location in patent:Paragraph 0179; 0180; 0181; 0182
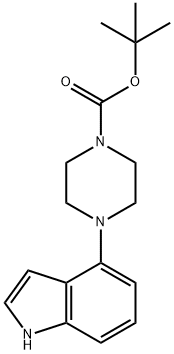
252978-89-5
52 suppliers
$28.00/100mg

84807-09-0
81 suppliers
$97.00/250mg
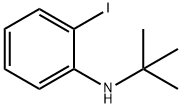
1562567-12-7
0 suppliers
inquiry

84807-09-0
81 suppliers
$97.00/250mg
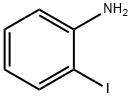
615-43-0
428 suppliers
$5.00/5g

84807-09-0
81 suppliers
$97.00/250mg