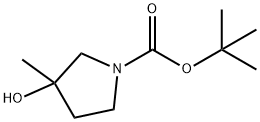
tert-Butyl 3-hydroxy-3-Methylpyrrolidine-1-carboxylate synthesis
- Product Name:tert-Butyl 3-hydroxy-3-Methylpyrrolidine-1-carboxylate
- CAS Number:412278-02-5
- Molecular formula:C10H19NO3
- Molecular Weight:201.26
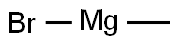
75-16-1
266 suppliers
$12.00/10ml
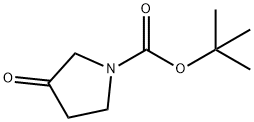
101385-93-7
471 suppliers
$5.00/1g

412278-02-5
52 suppliers
$65.00/100mg
Yield:412278-02-5 92%
Reaction Conditions:
in diethyl ether at 20; for 1 h;Inert atmosphere;
Steps:
tert-Butyl 3-hydroxy-3-methylpyrrolidine-1-carboxylate (i)
To an ice-cold solution of tert-butyl 3-oxopyrrolidine-1-carboxylate (1.0 g, 5.40 mmol) in Et2O (20 mL) was added methylmagnesium bromide (3.50 mL, 10.80 mmol.) drop wise under an inert atmosphere.
The mixture was stirred at RT for 1 h, then quenched with NH4Cl solution followed by extraction with EtOAc (3*100 mL).
The combined organic layers were washed with brine, dried (Na2SO4), filtered and concentrated in vacuo.
Trituration with n-pentane gave i (1.0 g, 92%).
3-Methylpyrrolidin-3-ol hydrochloride (Precursor 52): To a solution of 4M HCl in 1,4-dioxane (10 mL) at 0° C. was added i (0.850 g, 4.22 mmol) and the mixture stirred for 2 h at RT.
References:
US2013/252938,2013,A1 Location in patent:Paragraph 0514

101385-93-7
471 suppliers
$5.00/1g

74-88-4
342 suppliers
$15.00/10g

412278-02-5
52 suppliers
$65.00/100mg
917-64-6
145 suppliers
$45.00/10ml

101385-93-7
471 suppliers
$5.00/1g

412278-02-5
52 suppliers
$65.00/100mg