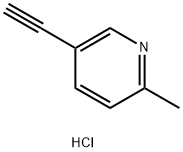
5-ethynyl-2-methylpyridine hydrochloride synthesis
- Product Name:5-ethynyl-2-methylpyridine hydrochloride
- CAS Number:1207351-11-8
- Molecular formula:C8H8ClN
- Molecular Weight:153.61
![Pyridine, 2-methyl-5-[2-(trimethylsilyl)ethynyl]-](/CAS/20210305/GIF/250252-33-6.gif)
250252-33-6
1 suppliers
inquiry

1207351-11-8
11 suppliers
inquiry
Yield:1207351-11-8 73%
Reaction Conditions:
with methanol;potassium carbonate at 20;
Steps:
A.A-13
General Procedure 3 (GP3): Desilylation of Alkynes; The TMS-alkyne (1.0 eq.) is dissolved in MeOH, K2CO3 (0.5 eq.) is added in one portion and the reaction mixture is stirred at RT until conversion is complete (3 - 16 h). The solvent is removed in vacuo, the crude product is dissolved in ethyl acetate and the organic phase is extracted with water. The organic phase is dried, filtered off and the solvent removed in vacuo. The product is either used without further purification or purified by chromatography on silica gel using a DCM/MeOH or (cyclo-)hexane/ethyl acetate.; A-13) 5-Ethynyl-2-methyl-pyridine; The title compound is synthesized according to general procedure GP3 starting from 2.2 g (12 mmol) 2-methyl-5-trimethylsilanylethynyl-pyridine (A4) and 0.80 g (5.8 mmol)K2CO3 in 13 mL MeOH. The crude product is purified by chromatography on silica gel using a cyclohexane/ethyl acetate gradient. The product is extracted from the organic phase with 1 N HCl and isolated as the hydrochloride after lyophilization. Yield: 1.3 g (73 %).
References:
WO2010/12740,2010,A1 Location in patent:Page/Page column 10; 17-18
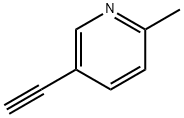
1945-85-3
23 suppliers
$45.00/10mg

1207351-11-8
11 suppliers
inquiry
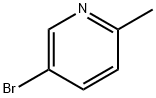
3430-13-5
429 suppliers
$5.00/5g

1207351-11-8
11 suppliers
inquiry