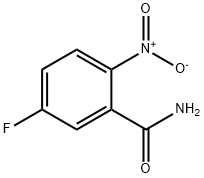
5-FLUORO-2-NITROBENZAMIDE synthesis
- Product Name:5-FLUORO-2-NITROBENZAMIDE
- CAS Number:77206-97-4
- Molecular formula:C7H5FN2O3
- Molecular Weight:184.12
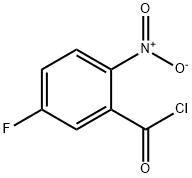
394-02-5

77206-97-4
General procedure for the synthesis of 5-fluoro-2-nitrobenzamide from 5-fluoro-2-nitrobenzoyl chloride: anhydrous tetrahydrofuran (THF, 3 mL) solution of oxalyl chloride (1.83 g, 14.4 mmol) was added slowly and dropwise at 0 °C and under nitrogen protection to commercially available 5-fluoro-2-nitrobenzoic acid (1.78 g, 9.62 mmol) and N,N-dimethylformamide ( DMF, 40 mg) in a solution of anhydrous THF (10 mL). After the reaction mixture was stirred at room temperature for 1.5 h, the solvent was removed by vacuum evaporation to give 5-fluoro-2-nitrobenzoyl chloride (2.13 g) as a yellow oil. Subsequently, a solution of anhydrous THF (9 mL) of 5-fluoro-2-nitrobenzoyl chloride (1.53 g) was slowly added to pre-cooled 35% ammonia (30 mL) at room temperature. The reaction mixture was stirred for 45 min and extracted by adding dichloromethane (50 mL). The organic layer was separated and the aqueous layer was extracted once more with dichloromethane (50 mL). The combined organic phases were washed with saturated aqueous ammonium chloride solution (1 x 50 mL), dried over anhydrous sodium sulfate and filtered, and the filtrate was concentrated to dryness in vacuum to afford 5-fluoro-2-nitrobenzamide (1.04 g, 75% yield, liquid chromatographic purity >95%). Mass spectrometry analysis: [M+H]+=185.
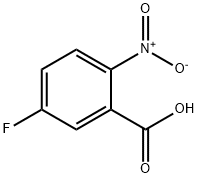
320-98-9
298 suppliers
$8.00/5g

77206-97-4
30 suppliers
inquiry
Yield:77206-97-4 96%
Reaction Conditions:
with ammonia;(benzotriazo-1-yloxy)tris(dimethylamino)phosphonium hexafluorophosphate;N-ethyl-N,N-diisopropylamine in 1,4-dioxane;dichloromethane at 20; for 16 h;Inert atmosphere;
Steps:
1.1 Step 1 :
Under inert atmosphere, a solution of 5-fluoro-2-nitrobenzoic acid (2.50 g, 13.5 mmol), ammonia (0.5M in dioxane, 54.0 ml_, 27.0 mmol), benzotriazol-1-yloxy- tris(dimethylamino)-phosphonium hexafluorophosphate (8.90 g, 20.3 mmol) and diisopropylethylamine (6.10 ml_, 35.1 mmol) in anhydrous dichloromethane (68.0 ml.) was stirred for 16 h at room temperature. Then, the mixture was poured into an aqueous saturated solution of ammonium chloride (250 ml.) and extracted with dichloromethane (2 x 200 ml_). The combined organic extracts were washed with brine (100 ml_), dried over MgS04 and concentrated under vacuum. The crude dark solid was purified by flash column chromatography on silica gel using cyclohexane / ethyl acetate as eluent to afford 5-fluoro-2-nitrobenzamide (2.40 g, 13.0 mmol, 96%) as a brown solid. (0777) M/Z (M+H)+ = 185.2.
References:
WO2020/21064,2020,A1 Location in patent:Page/Page column 103

394-02-5
31 suppliers
$13.00/250mg

77206-97-4
30 suppliers
inquiry
455-38-9
407 suppliers
$11.92/25g

77206-97-4
30 suppliers
inquiry