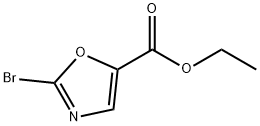
5-Oxazolecarboxylic acid, 2-bromo-, ethyl ester synthesis
- Product Name:5-Oxazolecarboxylic acid, 2-bromo-, ethyl ester
- CAS Number:1060816-22-9
- Molecular formula:C6H6BrNO3
- Molecular Weight:220.02
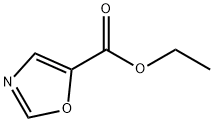
118994-89-1
183 suppliers
$20.00/1g

1060816-22-9
94 suppliers
$32.00/100mg
Yield:1060816-22-9 68%
Reaction Conditions:
Stage #1: oxazol-5-yl-carboxylic acid ethyl esterwith lithium hexamethyldisilazane in tetrahydrofuran at -80 - -70; for 0.166667 h;Inert atmosphere;
Stage #2: with carbon tetrabromide in tetrahydrofuran at -80 - -70; for 2 h;
Steps:
1 Example 1
Under a argon atmosphere, tetrahydrofuran (500 mL) was added to the reaction flask andCompound 1 is oxazole-5-carboxylic acid ethyl ester (32 g 0.23 mol 1 eq). The temperature was lowered to -70 to -80 ° C, and then LiHMDS (250 ml, 0.25 mol, 1.1 eq.) was slowly added dropwise.After the dropwise addition, the temperature was kept at -70 to -80 ° C, and the reaction was stirred for 10 minutes.CBr4 (106 g, 0.32 mol, 1.4 eq.) was added in portions, and after the addition, the temperature was kept at -70 to -80 ° C, and the reaction was stirred for 2 hours.After extraction with saturated ammonium chloride, it was slowly warmed to room temperature.Extract with ethyl acetate (250 mL * 2), wash with water (250 mL * 2), and wash with saturated brine (250 mL * 1).Concentrated and mixed with the column to obtain 34.4 g of product, the yield was 68%.
References:
CN109666005,2019,A Location in patent:Paragraph 0026; 0027; 0028
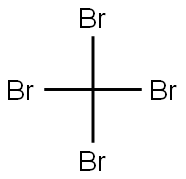
558-13-4
251 suppliers
$10.00/1g

118994-89-1
183 suppliers
$20.00/1g

1060816-22-9
94 suppliers
$32.00/100mg