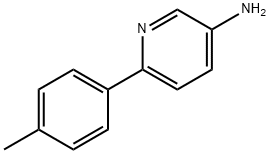
6-P-TOLYLPYRIDIN-3-YLAMINE synthesis
- Product Name:6-P-TOLYLPYRIDIN-3-YLAMINE
- CAS Number:170850-45-0
- Molecular formula:C12H12N2
- Molecular Weight:184.24

131941-26-9

170850-45-0
4.3.1 Synthesis of 4'-methyl-4-aminobiphenyl (4a): 5-nitro-2-(p-tolyl)pyridine (3a, 2.54 mmol, 541 mg) was dissolved in a solvent mixture of EtOH/EtOAc (1:1, 50 mL). The hydrogenation-reduction reaction was carried out in all-hydrogen mode in the presence of Raney Nickel catalyst at a reaction temperature of 65 °C and a hydrogen flow rate of 0.8 mL/min. Upon completion of the reaction, the resulting solution was concentrated under reduced pressure to give 480 mg of yellow solid. The crude product was purified by silica gel column chromatography using hexane/CH2Cl2 (1:1) as eluent, resulting in 440 mg of the pure target product 4'-methyl-4-aminobiphenyl (4a) as an off-white solid in 95% yield.

131941-26-9
3 suppliers
inquiry

170850-45-0
61 suppliers
$60.00/250mg
Yield: 99%
Reaction Conditions:
with nickel in ethanol;ethyl acetate at 65;
Steps:
3.2 General procedure for the nitro group reduction
General procedure: 4.3.1 4'-Methyl-4-aminobiphenyl (4a) Compound 3a (2.54 mmol, 541 mg) was dissolved in an EtOH/EtOAc 1:1 mixture (50 mL) and reduced by hydrogenation catalysed by Raney Nickel in an H-Cube at 65 °C at a flow rate of 0.8 mL/min, using full hydrogen mode. The solution obtained was concentrated under reduced pressure to give 480 mg of a yellow solid, which was purified by chromatography on silica gel, hexane/CH2Cl2 1:1, to give 440 mg of the pure expected product as an off-white solid (95% yield).
References:
Baltus, Christine B.;Press, Neil J.;Antonijevic, Milan D.;Tizzard, Graham J.;Coles, Simon J.;Spencer, John [Tetrahedron,2012,vol. 68,# 45,p. 9272 - 9277]
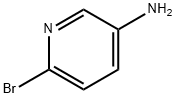
13534-97-9
437 suppliers
$6.00/1g
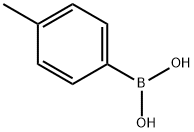
5720-05-8
439 suppliers
$5.00/1g

170850-45-0
61 suppliers
$60.00/250mg
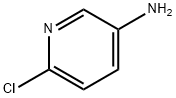
5350-93-6
430 suppliers
$6.00/5g

5720-05-8
439 suppliers
$5.00/1g

170850-45-0
61 suppliers
$60.00/250mg
4487-59-6
445 suppliers
$5.00/1g

170850-45-0
61 suppliers
$60.00/250mg

5720-05-8
439 suppliers
$5.00/1g

170850-45-0
61 suppliers
$60.00/250mg