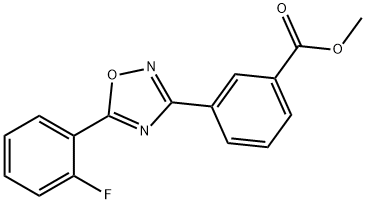
Benzoic acid, 3-[5-(2-fluorophenyl)-1,2,4-oxadiazol-3-yl]-, Methyl ester synthesis
- Product Name:Benzoic acid, 3-[5-(2-fluorophenyl)-1,2,4-oxadiazol-3-yl]-, Methyl ester
- CAS Number:775304-60-4
- Molecular formula:C16H11FN2O3
- Molecular Weight:298.27
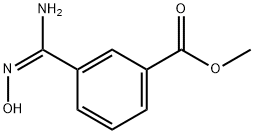
1141475-82-2
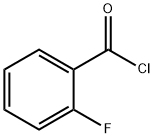
393-52-2
![Benzoic acid, 3-[5-(2-fluorophenyl)-1,2,4-oxadiazol-3-yl]-, Methyl ester](/CAS/GIF/775304-60-4.gif)
775304-60-4
GENERAL METHOD: 2 mmol of methyl (E)-3-(N'-hydroxycarbamoyl)benzoate was dissolved in 200 mL of acetone in a round-bottomed flask; subsequently, 0.35 g (2.5 mmol) of K2CO3 and 2.5 mmol of o-fluorobenzoyl chloride were added to the reaction mixture, and the reaction was stirred for 24 h at room temperature. Upon completion of the reaction, the solvent was removed by distillation under reduced pressure and the residue was treated with an appropriate amount of water and refluxed for 30 minutes. After the reaction mixture was cooled, the 1,2,4-oxadiazole product was collected by filtration and further purified by column chromatography to afford the target compound, methyl 3-(5-(2-fluorophenyl)-1,2,4-oxadiazol-3-yl)benzoate.
775304-80-8
8 suppliers
inquiry
![Benzoic acid, 3-[5-(2-fluorophenyl)-1,2,4-oxadiazol-3-yl]-, Methyl ester](/CAS/GIF/775304-60-4.gif)
775304-60-4
45 suppliers
$19.00/250mg
Yield:775304-60-4 92%
Reaction Conditions:
in toluene at 5 - 130; for 22 h;
Steps:
6.7
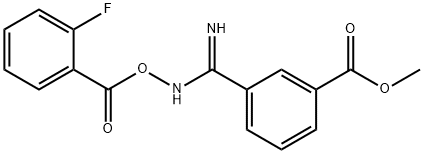
44g of 3-(N-2-Fluorobenzoylcarbamimidoyl)-benzoic acid methyl ester in toluene (500 mL) was refluxed for 4h at 130 °C using Dean-Stark apparatus. The reaction mixture was stirred at 5 °C for 18h. The white precipitate was filtered off and the filtrate was concentrated, crystallized again in toluene. The desired oxadiazole (38g, 92% yield) was obtained as a white solid with 99% purity (LC/UV). 1H-NMR (CDCl3) S 8.91 (IH)5 8.38 (IH)3 8.15 (2H), 7.62 (2H)3 7.35 (2H)3 3.95 (3H).
References:
WO2007/117438,2007,A2 Location in patent:Page/Page column 261
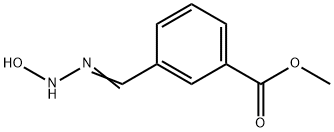
775304-79-5
11 suppliers
inquiry

393-52-2
396 suppliers
$10.00/25g
![Benzoic acid, 3-[5-(2-fluorophenyl)-1,2,4-oxadiazol-3-yl]-, Methyl ester](/CAS/GIF/775304-60-4.gif)
775304-60-4
45 suppliers
$19.00/250mg
![Benzoic acid, 3-[(hydroxyimino)methyl]-, methyl ester](/CAS/20180629/GIF/344412-41-5.gif)
344412-41-5
2 suppliers
inquiry
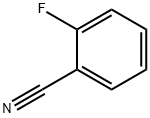
394-47-8
479 suppliers
$5.00/5g
![Benzoic acid, 3-[5-(2-fluorophenyl)-1,2,4-oxadiazol-3-yl]-, Methyl ester](/CAS/GIF/775304-60-4.gif)
775304-60-4
45 suppliers
$19.00/250mg
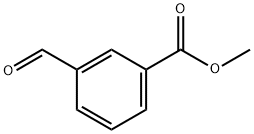
52178-50-4
174 suppliers
$22.00/1g
![Benzoic acid, 3-[5-(2-fluorophenyl)-1,2,4-oxadiazol-3-yl]-, Methyl ester](/CAS/GIF/775304-60-4.gif)
775304-60-4
45 suppliers
$19.00/250mg
67-56-1
790 suppliers
$9.00/25ml
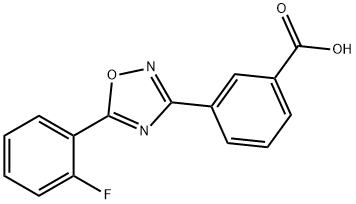
775304-57-9
242 suppliers
$6.00/5mg
![Benzoic acid, 3-[5-(2-fluorophenyl)-1,2,4-oxadiazol-3-yl]-, Methyl ester](/CAS/GIF/775304-60-4.gif)
775304-60-4
45 suppliers
$19.00/250mg