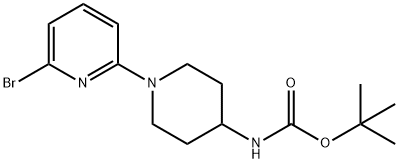
(6'-BroMo-3,4,5,6-tetrahydro-2H-[1,2']bipyridinyl-4-yl)-carbaMic acid tert-butyl ester, 98+% C15H22BrN3O2, MW: 356.27 synthesis
- Product Name:(6'-BroMo-3,4,5,6-tetrahydro-2H-[1,2']bipyridinyl-4-yl)-carbaMic acid tert-butyl ester, 98+% C15H22BrN3O2, MW: 356.27
- CAS Number:848500-12-9
- Molecular formula:C15H22BrN3O2
- Molecular Weight:356.26
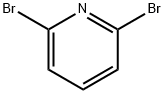
626-05-1
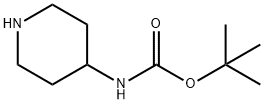
73874-95-0
![(6'-BroMo-3,4,5,6-tetrahydro-2H-[1,2']bipyridinyl-4-yl)-carbaMic acid tert-butyl ester, 98+% C15H22BrN3O2, MW: 356.27](/CAS/GIF/848500-12-9.gif)
848500-12-9
The reaction of 2,6-dibromopyridine (3.5 g, 14.77 mmol) and 4-tert-butoxycarbonylaminopiperidine (1.478 g, 7.38 mmol) was carried out in a sealed vessel at 130 °C for 6.5 hours, followed by warming up to 160 °C to continue the reaction for 3 hours. After completion of the reaction, the mixture was cooled to room temperature, dissolved in dichloromethane (CH2Cl2), washed sequentially with saturated sodium bicarbonate (NaHCO3) solution (twice) and brine, and the organic phase was dried with anhydrous sodium sulfate (Na2SO4), filtered and concentrated. The residue was purified by fast column chromatography (silica gel, 10-30% ethyl acetate/hexane gradient elution) to afford the intermediate (6'-bromo-3,4,5,6-tetrahydro-2H-[1,2']bipyridin-4-yl)-carbamic acid tert-butyl ester (0.83 g, 32% yield). Mass Spectra (ESI) m/z 356.0, 358.0 ([M+H]+) The intermediate was used directly in the next step of the reaction.

626-05-1
507 suppliers
$9.00/10g

73874-95-0
443 suppliers
$10.00/250mg
![(6'-BroMo-3,4,5,6-tetrahydro-2H-[1,2']bipyridinyl-4-yl)-carbaMic acid tert-butyl ester, 98+% C15H22BrN3O2, MW: 356.27](/CAS/GIF/848500-12-9.gif)
848500-12-9
26 suppliers
$195.00/250mg
Yield:848500-12-9 32%
Reaction Conditions:
at 130 - 160; for 9.5 h;
Steps:
64.A
A mixture of 2,6-dibromo-pyridine (3.5 g, 14.77 mmol) and piperidin-4-yl-carbamic acid te/f-butyl ester (1.478 g, 7.38 mmol) is heated to 130 0C in a sealed vessel for 6.5 h and 160 0C for 3 h. The residue is cooled and dissolved in CH2CI2 , washed with saturated NaHCO3 (X2), brine, dried (Na2SO4), filtered and concentrated. The residue is then separated via flash chromatography (SiO2, 10-30% EtOAc/hexanes gradient) to give an intermediate, (6'-bromo-3,4,5,6-tetrahydro-2H-[1 ,2']bipyridinyl-4-yl)-carbamic acid te/f-butyl ester (0.83 g, 32%). MS (ESI) m/z 356.0, 358.0 (M+1 ), which is taken on to next step.
References:
WO2009/150230,2009,A1 Location in patent:Page/Page column 179