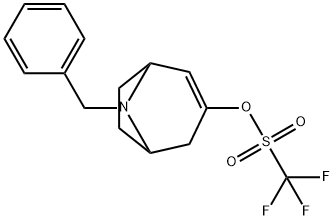
8-Benzyl-3-(trifluoroMethylsulfonyloxy)-8-azabicyclo[3.2.1]oct-3-ene synthesis
- Product Name:8-Benzyl-3-(trifluoroMethylsulfonyloxy)-8-azabicyclo[3.2.1]oct-3-ene
- CAS Number:949902-02-7
- Molecular formula:C15H16F3NO3S
- Molecular Weight:347.35
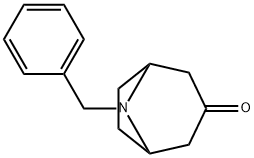
28957-72-4
208 suppliers
$28.00/1g
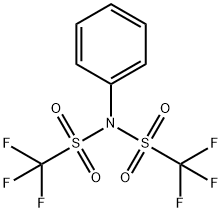
![2-[N,N-BIS(TRIFLUOROMETHANESULFONYL)AMINO]-5-CHLOROPYRIDINE](/CAS/GIF/145100-51-2.gif)
145100-51-2
217 suppliers
$6.00/1g
![8-Benzyl-3-(trifluoroMethylsulfonyloxy)-8-azabicyclo[3.2.1]oct-3-ene](/CAS/20150408/GIF/949902-02-7.gif)
949902-02-7
11 suppliers
inquiry
Yield:949902-02-7 62%
Reaction Conditions:
Stage #1: N-benzyltropinonewith lithium hexamethyldisilazane in tetrahydrofuran at -78; for 1 h;
Stage #2: N-(5-chloropyridin-2-yl)-1,1,1-trifluoro-N-[(trifluoromethyl)sulphonyl]methanesulphonamide in tetrahydrofuran at -78 - 20;
Steps:
IV
Lithium bis(trimethylsilyl)amide (1 mol/L in tetrahydrofuran, 51.5 ml.) is added to a solution of 8-benzyl-8-aza-bicyclo[3.2.1]octan-3-one (10.6 g) in tetrahydrofuran (200 ml.) cooled to -78 0C. The solution is stirred at -78 0C for 1 h, before 2-(N,N-(bistrifluoromethylsulfonyl)amino)- 5-chloropyridine (20.8 g) dissolved in tetrahydrofuran (200 mL) is added dropwise. The resulting solution is stirred for another 0.5 h at this temperature and then warmed to room temperature by removing the cooling bath. Then, aqueous NaHCC>3 solution is added, the resulting mixture is extracted with ethyl acetate, and the combined extracts are dried (Na2SC>4). After removal of the solvent under reduced pressure, the residue is purified by chromatography on silica gel (cyclohexane/ethyl acetate 1 :0->1 :1 ). Yield: 10.5 g (62% of theory)Mass spectrum (ESI+): m/z = 348 [M+H]+
References:
WO2010/46445,2010,A2 Location in patent:Page/Page column 56-57
37595-74-7
427 suppliers
$5.00/1g
![8-Benzyl-3-(trifluoroMethylsulfonyloxy)-8-azabicyclo[3.2.1]oct-3-ene](/CAS/20150408/GIF/949902-02-7.gif)
949902-02-7
11 suppliers
inquiry

37595-74-7
427 suppliers
$5.00/1g
![8-(Phenylmethyl)-8-azabicyclo[3.2.1]octan-3-one hydrochloride](/CAS2/GIF/83393-23-1.gif)
83393-23-1
55 suppliers
$35.00/1g
![8-Benzyl-3-(trifluoroMethylsulfonyloxy)-8-azabicyclo[3.2.1]oct-3-ene](/CAS/20150408/GIF/949902-02-7.gif)
949902-02-7
11 suppliers
inquiry