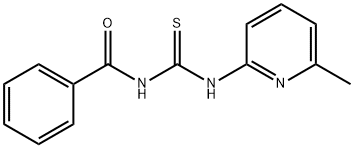
Benzamide, N-[[(6-methyl-2-pyridinyl)amino]thioxomethyl]- synthesis
- Product Name:Benzamide, N-[[(6-methyl-2-pyridinyl)amino]thioxomethyl]-
- CAS Number:96938-51-1
- Molecular formula:C14H13N3OS
- Molecular Weight:271.34
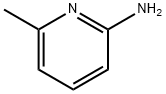
1824-81-3
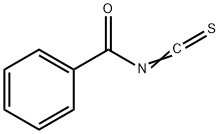
532-55-8
![Benzamide, N-[[(6-methyl-2-pyridinyl)amino]thioxomethyl]-](/CAS/20180629/GIF/96938-51-1.gif)
96938-51-1
To a solution of benzoyl isothiocyanate (83.0 g, 509.3 mmol) in acetone (700 mL) was slowly added a solution of 6-methylpyridin-2-amine (50 g, 463.0 mmol) in acetone (600 mL) dropwise. The reaction mixture was heated to reflux and stirred for 3 hours. Upon completion of the reaction, the mixture was decanted onto crushed ice, and the resulting precipitate was filtered and washed sequentially with water, water/methanol (1:1), and methanol to afford 1-benzoyl-3-(6-methylpyridin-2-yl)thiourea as a yellow solid (100.1 g, 80% yield). 1-Benzoyl-3-(6-methylpyridin-2-yl)thiourea (60 g, 221.4 mmol) was dissolved in THF (1000 mL), 2N NaOH solution (243.5 mL) was added and heated to reflux for 3 hours. After cooling to room temperature and filtering, (6-methylpyridin-2-yl)thiourea was obtained as a white solid (34.1 g, 92% yield). (6-Methylpyridin-2-yl)thiourea (13.2 g, 79.16 mmol) was mixed with 2-bromo-1-(4-bromophenyl)ethan-1-one (22 g, 79.16 mmol) in ethanol (300 mL) and heated to reflux and stirred for 3 hours. The reaction solution was concentrated and purified by silica gel column chromatography to afford N-[4-(4-bromophenyl)-1,3-thiazol-2-yl]-6-methylpyridin-2-amine as a yellow solid (14.3 g, 53% yield). N-[4-(4-bromophenyl)-1,3-thiazol-2-yl]-6-methylpyridin-2-amine (5 g, 14.5 mmol), bis(pinacolato)diboron (4.8 g, 18.8 mmol), Pd(dppf)2Cl2 (1.2 mg, 1.5 mmol) and AcOK (4.3 g, 43.3 mmol) were dissolved in dioxane ( 100 mL) and heated to 80 °C for overnight reaction. Evaporation of the solvent gave 6-methyl-N-{4-[4-(tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl]-1,3-thiazol-2-yl}pyridin-2-amine. The coupling reaction with 2-dimethylamino-4-bromopyridine (200 mg) was carried out under standard conditions to give 33 mg as a yellow solid (17% yield).

1824-81-3
487 suppliers
$8.00/25g

532-55-8
216 suppliers
$9.00/1g
![Benzamide, N-[[(6-methyl-2-pyridinyl)amino]thioxomethyl]-](/CAS/20180629/GIF/96938-51-1.gif)
96938-51-1
24 suppliers
$20.00/100mg
Yield:96938-51-1 80%
Reaction Conditions:
in acetone; for 3 h;Reflux;
Steps:
N,N-dimethyl-4-(4-{2-[(6-methylpyridin-2-yl)amino]-1,3-thiazol-4-yl}phenyl)pyridin-2-amine
To the solution of benzylisothiocyanate (83.0 g, 509.3 mmol) in acetone (700mL) was added compound 6-methylpyridin-2 -amine (50 g, 463.0mmol) in acetone (600 ml) dropwise, then the reaction mixture was stirred at reflux for 3h. The reaction mixture was poured on to crushed ice, then filtered and washed with water, water/MeOH (1 : 1) and MeOH to give l-benzoyl-3-(6- methylpyridin-2-yl)thiourea as a yellow solid (100.1 g, yield 80%). To a solution of 1-benzoyl-3-(6-methylpyridin-2-yl)thiourea (60 g,221.4 mmol) in THF ( 1000 ml) was added 2N NaOH (243.5ml), then heated at reflux for 3 h. Cooled to RT and filtered to give (6-methylpyridin-2- yl)thiourea as a white solid (34.1 g, yield 92%). A mixture of (6-methylpyridin-2-yl)thiourea (13.2 g, 79.16 mmol) and 2-bromo-l-(4-bromophenyl)ethan-l-one (22 g, 79.16 mmol) in ethanol (300 mL) was stirred at reflux for 3h, then concentrated and purified with silica gel column to give N-[4-(4-bromophenyl)-l,3-thiazol-2-yl]-6-methylpyridin-2-amine as a yellow solid (14.3 g,53%). A solution of N-[4-(4-bromophenyl)-l,3-thiazol-2-yl]-6-methylpyridin-2-amine (5 g. 14.5mmol), Bis(pinacolato)diboron (4.8 g, 18.8mmol), Pd(dppf)2Ci2 (1.2 mg, 1.5mmol) and AcOK (4.3 g, 43.3mmol) in dioxane (100ml) was heated to 80 under 2 overnight. The mixture was evaporated to give (6-methyl-N-{4-[4-(tetramethyl-l,3,2-dioxaborolan-2-yl)phenyl]-l,3-thiazol-2-yl}pyridin-2-amine). Coupling (200mg) with 2-dimethylamino-4- bromopyridine under standard conditions gave 33 mg from 200 mg of, yellow solid, 17 % yield.
References:
WO2013/33037,2013,A2 Location in patent:Paragraph 0752

1824-81-3
487 suppliers
$8.00/25g
333-20-0
462 suppliers
$13.50/100G
98-88-4
597 suppliers
$10.00/5g
![Benzamide, N-[[(6-methyl-2-pyridinyl)amino]thioxomethyl]-](/CAS/20180629/GIF/96938-51-1.gif)
96938-51-1
24 suppliers
$20.00/100mg
65-85-0
1437 suppliers
$10.00/25g
![Benzamide, N-[[(6-methyl-2-pyridinyl)amino]thioxomethyl]-](/CAS/20180629/GIF/96938-51-1.gif)
96938-51-1
24 suppliers
$20.00/100mg