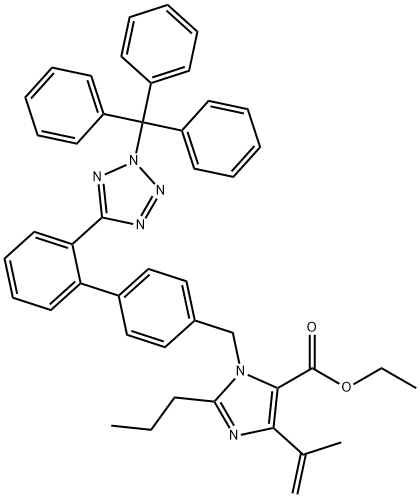
Dehydro N2-TriphenylMethyl OlMesartan Ethyl Ester synthesis
- Product Name:Dehydro N2-TriphenylMethyl OlMesartan Ethyl Ester
- CAS Number:172875-70-6
- Molecular formula:C45H42N6O2
- Molecular Weight:698.85
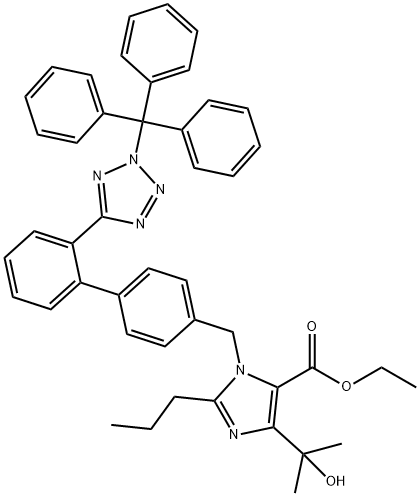
172875-59-1
51 suppliers
inquiry

172875-70-6
12 suppliers
$1320.00/5g
Yield:172875-70-6 14.2 g
Reaction Conditions:
with pyridine;trichlorophosphate in toluene at 0; for 1 h;Reflux;
Steps:
2
21.5 g of a compound of formula 1 was added to a 1 L reaction flask,Then add 200ml of toluene,After the addition was completed, the reaction solution was heated to reflux until all of Formula 1 was dissolved, and then the system was cooled to room temperature.Use an ice-water bath to reduce the temperature to 0 ° C, then add 5 ml of pyridine, and maintain the temperature of 0 ° C-5 ° C, and dropwise add a solution of phosphorus oxychloride (1.5 g of phosphorus oxychloride in 15 ml of toluene)After the addition is complete, heat the reaction to reflux for 1 hour(TLC detects that the reaction of Formula 1 is complete).Then the reaction solution was cooled to room temperature and added to 200 g of ice. The layers were stirred and separated. The organic layer was washed with saturated brine to neutrality, and the organic layer was concentrated in toluene.The residual liquid column chromatography was used to purify and collect the eluent rich in formula 4.The solvent was distilled off under reduced pressure to obtain 14.2 g of a reference product of Formula 4 with an HPLC purity of 99.36% (see Figure 1).
References:
CN110452228,2019,A Location in patent:Paragraph 0103-0113
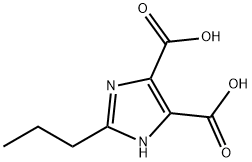
58954-23-7
161 suppliers
$100.00/50mg

172875-70-6
12 suppliers
$1320.00/5g
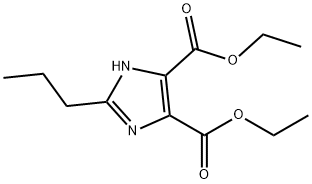
144689-94-1
279 suppliers
$8.00/5g

172875-70-6
12 suppliers
$1320.00/5g
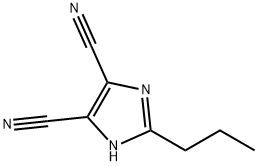
51802-42-7
79 suppliers
inquiry

172875-70-6
12 suppliers
$1320.00/5g
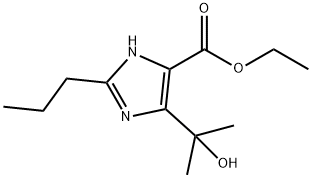
144689-93-0
488 suppliers
$16.00/5g

172875-70-6
12 suppliers
$1320.00/5g