
ETHYL (4-AMINOPHENYLAMINO) OXOACETATE synthesis
- Product Name:ETHYL (4-AMINOPHENYLAMINO) OXOACETATE
- CAS Number:17794-28-4
- Molecular formula:C10H12N2O3
- Molecular Weight:208.21

5416-11-5
17 suppliers
inquiry

17794-28-4
12 suppliers
$155.00/1g
Yield:17794-28-4 92%
Reaction Conditions:
with palladium on activated charcoal;hydrogen in tetrahydrofuran at 20; for 3 h;
Steps:
4.7.2. General procedure A. Synthesis of compounds 4a-c (with 4a as an example)
General procedure: General procedure A.
Synthesis of compounds 4a-c (with 4a as an example)
Compound 3a (1.00 g, 4.20 mmol) was dissolved in tetrahydrofuran (50 mL), Pd-C (200 mg) was added and the reaction mixture was stirred under hydrogen atmosphere for 3 h.
The catalyst was filtered off and the solvent removed under reduced pressure to give 4a (0.80 g) as a brown solid. 4.7.3
Ethyl 2-((4-aminophenyl)amino)-2-oxoacetate (4a)
Brown solid; yield 92% (0.80 g); mp 110-112 °C (110-113 °C, lit
[38]
); IR (ATR) ν = 3476, 3317, 3348, 3217, 2981, 2931, 1728, 1672, 1516, 1282, 1177, 824 cm-1. 1H NMR (400 MHz, DMSO-d6) δ 1.30 (t, 3H, 3J = 7.2 Hz, CH3), 4.25-4.31 (m, 2H, CH2), 5.06 (s, 2H, NH2), 6.52 (d, 2H, 3J = 8.8 Hz, Ar-H-2,6), 7.37 (d, 2H, 3J = 8.8 Hz, Ar-H-3,5), 10.39 (s, 1H, NH).
References:
Zidar, Nace;Toma?i?, Tihomir;Macut, Helena;Sirc, Anja;Brvar, Matja?;Montalv?o, Sofia;Tammela, P?ivi;Ila?, Janez;Kikelj, Danijel [European Journal of Medicinal Chemistry,2016,vol. 117,p. 197 - 211] Location in patent:supporting information
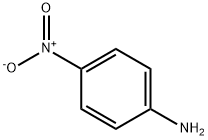
100-01-6
10 suppliers
$11.19/25G

17794-28-4
12 suppliers
$155.00/1g