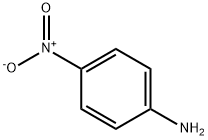
4-Nitroaniline
- CAS No.
- 100-01-6
- Chemical Name:
- 4-Nitroaniline
- Synonyms
- PNA;P-NITROANILINE;PARA NITRO ANILINE;4-NITROBENZENAMINE;P-nitroanilide;4-Nitro-phenylamine;P-NITROPHENYLAMINE;red2gbase;developerp;devolredgg
- CBNumber:
- CB9852549
- Molecular Formula:
- C6H6N2O2
- Molecular Weight:
- 138.12
- MDL Number:
- MFCD00007858
- MOL File:
- 100-01-6.mol
- MSDS File:
- SDS
| Melting point | 147 °C |
|---|---|
| Boiling point | 332 °C |
| Density | 1,437 g/cm3 |
| vapor density | 4.77 (vs air) |
| vapor pressure | 1.33 hPa (142 °C) |
| refractive index | 1.6465 (estimate) |
| Flash point | 329 °F |
| storage temp. | Store below +30°C. |
| solubility | ethanol: soluble25mg/mL, slightly hazy, yellow to brownish-yellow |
| pka | 1(at 25℃) |
| Colour Index | 37035 |
| form | crystalline |
| color | yellow |
| Specific Gravity | 1.437 |
| Odor | Ammonia odor |
| PH Range | 7 |
| Water Solubility | 0.8 g/L (20 ºC) |
| Merck | 14,6584 |
| BRN | 508690 |
| Henry's Law Constant | 1.15 x 10-9 atm?m3/mol at 25 °C (thermodynamic method-GC/UV spectrophotometry, Altschuh et al., 1999) |
| Exposure limits | NIOSH REL: TWA 3; IDLH 300; OSHA PEL: TWA 6; ACGIH TLV: TWA 3 (adopted). |
| Dielectric constant | 56.3(160.0℃) |
| Stability | Stable. Incompatible with sodium hydroxide, strong oxidizing agents, strong reducing agents. May attack some plastics, rubber and coatings. May decompose explosively in the presence of initiators. Moisture sensitive. May be light and air-sensitive. |
| LogP | 1.2 at 30℃ |
| CAS DataBase Reference | 100-01-6(CAS DataBase Reference) |
| EWG's Food Scores | 2 |
| FDA UNII | 1MRQ0QZG7G |
| NIST Chemistry Reference | p-Nitroaniline(100-01-6) |
| EPA Substance Registry System | p-Nitroaniline (100-01-6) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |   GHS06,GHS08 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H301+H311+H331-H373-H412 | |||||||||
| Precautionary statements | P273-P280-P301+P310-P302+P352+P312-P304+P340+P311-P314 | |||||||||
| Hazard Codes | T,F | |||||||||
| Risk Statements | 23/24/25-33-52/53-39/23/24/25-11 | |||||||||
| Safety Statements | 28-36/37-45-61-28A-16-1/2-7 | |||||||||
| RIDADR | UN 1661 6.1/PG 2 | |||||||||
| OEB | B | |||||||||
| OEL | TWA: 3 mg/m3 [skin] | |||||||||
| WGK Germany | 2 | |||||||||
| RTECS | BY7000000 | |||||||||
| F | 8 | |||||||||
| Autoignition Temperature | 500 °C | |||||||||
| Hazard Note | Toxic | |||||||||
| TSCA | Yes | |||||||||
| HazardClass | 6.1 | |||||||||
| PackingGroup | II | |||||||||
| HS Code | 29214210 | |||||||||
| Toxicity | Acute LD50 for wild birds 75 mg/kg, guinea pigs 450 mg/kg, mice 810 mg/kg, quail 1,000 mg/kg, rats 750 mg/kg (quoted, RTECS, 1985). | |||||||||
| IDLA | 300 mg/m3 | |||||||||
| NFPA 704 |
|
4-Nitroaniline Chemical Properties,Uses,Production
Chemical Properties
p-Nitroaniline consists of yellow crystals with a pungent, faint ammonia-like odor.
Chemical Properties
Yellow fine crystals
Physical properties
Bright yellow crystalline powder or flakes with a faint, ammonia-like, slightly pungent odor. Combustible.
Uses
Product of chromogenic reactions.
Uses
Dyestuff intermediate.
Uses
4-Nitroaniline is used in the synthesis of photorefractive polymers, as novel chromophores in analytical study. As well, due to the absorption spectrum associated with 4-Nitroaniline, it is used to de termine catechol derivatives in syntheses.
Definition
ChEBI: A nitroaniline carrying a nitro group at position 4.
Synthesis Reference(s)
The Journal of Organic Chemistry, 59, p. 4114, 1994 DOI: 10.1021/jo00094a021
Synthetic Communications, 20, p. 293, 1990 DOI: 10.1080/00397919008052297
General Description
Yellow solid with a mild odor. Sinks in water.
Air & Water Reactions
4-Nitroaniline may be sensitive to prolonged exposure to air and light. Insoluble in water. 4-Nitroaniline is sensitive to moisture.
Reactivity Profile
P-NITROANILINE may react vigorously with sulfuric acid above 392° F. 4-Nitroaniline may also react with sodium hydroxide at 266° F. Under pressure, 4-Nitroaniline may produce an explosive compound. 4-Nitroaniline is incompatible with strong oxidizers and strong reducing agents. 4-Nitroaniline is capable of explosive decomposition with strong initiators. 4-Nitroaniline will attack some forms of plastics, rubber and coatings.
Hazard
Explosion risk. Toxic when absorbed by skin. Methemoglobinemia, liver damage and eye irritant. Questionable carcinogen.
Health Hazard
Inhalation or ingestion causes headache, drowsiness, shortness of breath, nausea, methemoglobinemia, and unconsciousness; fingernails, lips, and ears become bluish; prolonged and excessive exposures may also cause liver damage. Contact with eyes causes irritation and possible corneal damage. Contact with skin causes irritation; continued exposure may cause same symptoms as inhalation or ingestion.
Flammability and Explosibility
Non flammable
Safety Profile
Poison by ingestion, intravenous, and intraperitoneal routes. Moderately toxic by intramuscular route. Mutation data reported. Acute symptoms of exposure are headache, nausea, vomiting, weakness and stupor, cyanosis and methemoglobinemia. Chronic exposure can cause liver damage. Experimental reproductive effects. Combustible when exposed to heat or flame. See NITRATES for explosion and disaster hazards. To fight fire, use water spray or mist, foam, dry chemical, CO2. Vigorous reaction with sulfuric acid above 200°C. Reaction with sodium hydroxide at 13O°C under pressure may produce the explosive sodium-4-nitrophenoxide. When heated to decomposition it emits toxic fumes of NOx. See also m- NITROANILINE, o-NITROANILINE, NITRO COMPOUNDS OF AROMATIC HYDROCARBONS, and ANILINE DYES.
Potential Exposure
p-Nitroaniline is used as an intermediate in the manufacture of dyes; antioxidants, pharmaceuticals, antiozonants, colors, pigments and pesticides.
Carcinogenicity
p-Nitroaniline has been tested in several strains of S. typhimurium. It was mutagenic in strain TA98 but not in strains TA97, TA100, TA1535, and TA1537, with and without S9. It caused gene mutation in mouse lymphoma cells and chromosomal aberrations in CHO cells in the presence of S9.
Environmental Fate
Biological. A Pseudomonas sp. strain P6, isolated from a Matapeake silt loam, was grown using
a yeast extract. After 8 d, 4-nitroaniline degraded completely to carbon dioxide (Zeyer and
Kearney, 1983). In activated sludge inoculum, following a 20-d adaptation period, no degradation
was observed (Pitter, 1976).
Chemical/Physical: Spacek et al. (1995) investigated the photodegradation of 4-nitroaniline
using titanium dioxide-UV light and Fenton’s reagent (hydrogen peroxide:substance – 10:1; Fe2+
2.5 x 10-4 mol/L). Both experiments were conducted at 25 °C. The decomposition rate of 4-nitroaniline
was very high by the photo-Fenton reaction in comparison to titanium dioxide-UV light (λ
= 365 nm). Decomposition products identified in both reactions were nitrobenzene, pbenzoquinone,
hydroquinone, oxalic acid, and resorcinol. Oxalic acid, hydroquinone, and pbenzoquinone
were identified as intermediate products using HPLC.
At influent concentrations of 10, 1.0, 0.1, and 0.01 mg/L, the GAC adsorption capacities were 250,
140, 74, and 40 mg/g, respectively (Dobbs and Cohen, 1980).
Shipping
UN1661 Nitroanilines (m-, o-, p-), Hazard Class: 6.1; Labels: 6.1-Poisonous materials.
Purification Methods
It also crystallises from acetone. It is freed from o-and m-isomers by zone melting and sublimation. [Beilstein 12 IV 1613.]
Incompatibilities
A combustible liquid. A strong oxidizer. Incompatible with strong acids; sulfur, combustibles, organics, and other easily oxidizable materials. Will accelerate the burning of combustible materials. If large quantities are involved in a fire or the combustible material is finely divided, an explosion may result. Prolonged exposure to fire or heat may result in an explosion.
Waste Disposal
Incineration (982℃, 2.0 seconds minimum) with scrubbing for nitrogen oxides abatement. Consult with environmental regulatory agencies for guidance on acceptable disposal practices. Generators of waste containing this contaminant (≥100 kg/mo) must conform with EPA regulations governing storage, transportation, treatment, and waste disposal.
4-Nitroaniline Preparation Products And Raw materials
Raw materials
Preparation Products
1of8