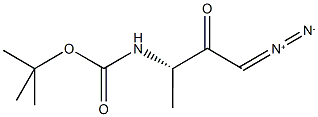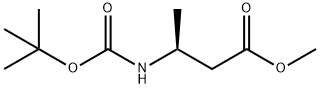
METHYL (S)-3-BOC-AMINOBUTYRATE synthesis
- Product Name:METHYL (S)-3-BOC-AMINOBUTYRATE
- CAS Number:106539-14-4
- Molecular formula:C10H19NO4
- Molecular Weight:217.26
67-56-1
787 suppliers
$9.00/25ml
158851-30-0
184 suppliers
$11.00/100mg
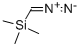
18107-18-1
209 suppliers
$33.00/5g

106539-14-4
11 suppliers
inquiry
Yield:106539-14-4 99%
Reaction Conditions:
at 20; for 18 h;
Steps:
146.1
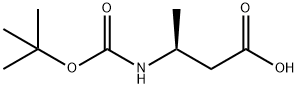
Example 146.2-Cyclopropyl-5H-pyrrolo[2,3-b]pyrazine-7-carboxylic acid ((S)-3-methanesulfonyl-l ,2,2- trimethyl-propyl) amideStep 1(S)-3-(tert-Butoxycarbonylamino)butanoic acid (1.0 g, 4.9 mmol) was dissolved in toluene (38 mL) and methanol (11 mL). The solution was cooled in an ice/water bath andtrimethylsilyl diazomethane (2 M solution in hexanes, 12.3 mL, 24.6 mmol) was added slowly. The reaction was stirred at 20°C for 18 h then concentrated. The residue was absorbed on to silica gel and purified by silica gel chromatography (ethyl acetate/hexanes) to give 1.06 g (99%) of (S)-3-tert-butoxycarbonylamino-butyric acid methyl ester.
References:
WO2011/144585,2011,A1 Location in patent:Page/Page column 214

158851-30-0
184 suppliers
$11.00/100mg

74-88-4
357 suppliers
$15.00/10g

106539-14-4
11 suppliers
inquiry
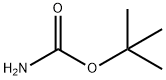
4248-19-5
376 suppliers
$5.00/5g

106539-14-4
11 suppliers
inquiry
![Carbamic acid, [(1S)-1-methyl-2-propenyl]-, 1,1-dimethylethyl ester (9CI)](/CAS/GIF/115378-33-1.gif)
115378-33-1
6 suppliers
inquiry

106539-14-4
11 suppliers
inquiry