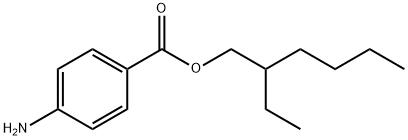
p-Aminobenzoesure-2-ethylhexylester synthesis
- Product Name:p-Aminobenzoesure-2-ethylhexylester
- CAS Number:26218-04-2
- Molecular formula:C15H23NO2
- Molecular Weight:249.35

16397-70-9

26218-04-2
The general procedure for the synthesis of 2-ethylhexyl 4-aminobenzoate from 2-ethylhexyl 4-nitrobenzoate was as follows: in a three-necked flask equipped with a stirrer, 920 mL of methanol-water solution (V methanol: V water = 5:1), 48.15 g (1.8 mol) of ammonium chloride, 33.60 g (1.2 mol) of reduced iron powder and 55.87 g (0.20 mol) 2-ethylhexyl 4-nitrobenzoate. The mixture was heated to reflux and the reaction was maintained for 16-17 hours. The progress of the reaction was monitored by thin layer chromatography (TLC) during the reaction. Upon completion of the reaction, the reaction mixture was filtered while hot and the diatomaceous earth filter cake was washed twice with methanol. The filtrate and washings were combined and concentrated under reduced pressure to obtain the crude product. The crude product was dissolved in 200 mL of water, the pH was adjusted to 7-8 with saturated sodium carbonate solution, and then extracted twice with 200 mL of dichloromethane. The organic phases were combined and dried by adding anhydrous magnesium sulfate for 3 hours. After drying, the magnesium sulfate was removed by filtration, and the filtrate was concentrated under reduced pressure to obtain the intermediate 2-ethylhexyl 4-aminobenzoate as a light yellow solid with 88.7% yield.

16397-70-9
50 suppliers
inquiry

26218-04-2
135 suppliers
$6.00/250mg
Yield:26218-04-2 99.2%
Reaction Conditions:
with palladium on activated charcoal;hydrogen in water at 50 - 60; under 750.075 - 3000.3 Torr;Sealed tube;
Steps:
1 Hydrogenation:
Put 619.8g of isooctyl p-nitrobenzoate in isooctyl alcohol into a dry and clean 2L hydrogenation kettle,2.1gPd/C (~5%, water content~60%), and 223.1g isooctyl alcohol;At the end of feeding, seal the hydrogenation kettle, replace it with nitrogen 3 times, and replace it 3 times with hydrogen; open the hydrogenation kettle and stir, set the hydrogen pressure in the kettle to 0.1-0.4Mpa, and the temperature of the kettle to 50-60°C. The hydrogenation reaction was started. During the reaction, the temperature of the kettle was maintained at 50-60°C. When the reaction did not absorb hydrogen any more, samples were taken and sent to GC for detection. When the isooctyl p-nitrobenzoate <0.1%, the reaction was stopped. Close the hydrogen main valve, lower the temperature of the kettle to 30-40°C, open the drain valve of the hydrogenation kettle, discharge the pressure in the kettle to 0.02Mpa, and replace the hydrogen in the kettle with nitrogen 3 times. The discharge port of the hydrogenation kettle was opened, and the reaction liquid was drawn out and filtered to obtain 2.3 g of catalyst and 847.3 g of filtrate. The catalyst was used in batch hydrogenation, and the filtrate was distilled under reduced pressure to obtain 401.3 g of isooctyl alcohol and 370.02 g of hydrogenation product isooctyl p-aminobenzoate. The GC purity was 99.56% and the yield was 99.2% (calculated as p-nitrobenzoic acid).
References:
CN112321522, 2021, A Location in patent:Paragraph 0071; 0095; 0097
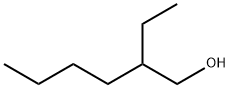
104-76-7
687 suppliers
$5.00/10g
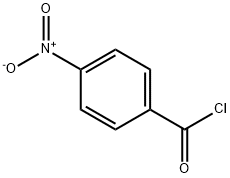
122-04-3
38 suppliers
$15.00/1g

26218-04-2
135 suppliers
$6.00/250mg
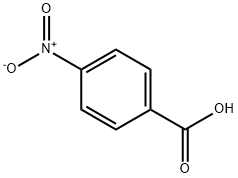
62-23-7
738 suppliers
$14.00/25g

26218-04-2
135 suppliers
$6.00/250mg

104-76-7
687 suppliers
$5.00/10g

26218-04-2
135 suppliers
$6.00/250mg