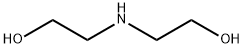
다이에탄올아민
|
|
다이에탄올아민 속성
- 녹는점
- 28 °C (lit.)
- 끓는 점
- 217 °C/150 mmHg (lit.)
- 밀도
- 1.097 g/mL at 25 °C (lit.)
- 증기 밀도
- 3.6 (vs air)
- 증기압
- <0.98 atm ( 100 °C)
- 굴절률
- n
20/D 1.477(lit.)
- 인화점
- 280 °F
- 저장 조건
- 2-8°C
- 용해도
- H2O: 1 M at 20 °C, 투명, 무색
- 물리적 상태
- 점성 액체 또는 저융점 고체
- 색상
- APHA: ≤15
- Specific Gravity
- 1.09
- 수소이온지수(pH)
- 11.0-12.0 (25℃, 1M in H2O)
- 산도 계수 (pKa)
- 8.88(at 25℃)
- 냄새
- 약한 암모니아성; 희미하고 비린내 나는; 특성.
- 폭발한계
- 2.1-10.6%(V)
- 수용성
- 혼용 가능
- 감도
- Hygroscopic
- 최대 파장(λmax)
- λ: 260 nm Amax: 0.04
λ: 280 nm Amax: 0.02
- Merck
- 14,3107
- BRN
- 605315
- 노출 한도
- TLV-TWA 3 ppm (~13 mg/m3) (ACGIH).
- Dielectric constant
- 2.8(25℃)
- 안정성
- 안정적인. 이산화탄소, 강산, 강산화제와 호환되지 않습니다. 조해성.
- InChIKey
- ZBCBWPMODOFKDW-UHFFFAOYSA-N
- LogP
- -2.46 at 25℃
- CAS 데이터베이스
- 111-42-2(CAS DataBase Reference)
- IARC
- 2B (Vol. 77, 101) 2013
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| 위험품 표기 | Xn | ||
|---|---|---|---|
| 위험 카페고리 넘버 | 22-38-41-48/22 | ||
| 안전지침서 | 26-36/37/39-46 | ||
| 유엔번호(UN No.) | 3267 | ||
| WGK 독일 | 1 | ||
| RTECS 번호 | KL2975000 | ||
| F 고인화성물질 | 3 | ||
| 자연 발화 온도 | 689 °F | ||
| TSCA | Yes | ||
| 포장분류 | II | ||
| HS 번호 | 29221200 | ||
| 유해 물질 데이터 | 111-42-2(Hazardous Substances Data) | ||
| 독성 | LD50 orally in rats: 12.76 g/kg (Smyth) | ||
| 기존화학 물질 | KE-20959 |
다이에탄올아민 C화학적 특성, 용도, 생산
개요
Diethanolamine is an organic base which has been used as an emulsifying and dispersing agent.It can also be used as a basic buffer, with optimal pH about pH 9, if titrated with HCl or other acid. Other uses include: to "scrub" gases, as a chemical intermediate, as humectant or softening agent.화학적 성질
The USP32–NF27 describes diethanolamine as a mixture of ethanolamines consisting largely of diethanolamine. At about room temperature it is a white, deliquescent solid. Above room temperature diethanolamine is a clear, viscous liquid with a mildly ammoniacal odor.
Diethanolamine is used as surface-active agent in metal-cutting fluids and oils, as a corrosion inhibitor, as a dispersant in agricultural chemical formulations, and as an intermediate in the production of other compounds such as fatty acid condensates of diethanolamine which are extensively used in soaps and cosmetics as emulsifiers, thickeners, wetting agents and detergents (Beyer et al., 1983). In the cosmetic formulations, the concentration of diethanolamine may range from 1 to 25% (National Toxicology Program, 1999a).
용도
Diethanolamine similar to triethanolamine (T775580) is used as a surfactant. It also has the potential to be a corrosion inhibitor by means of chemisorption.제조 방법
Diethanolamine is prepared commercially by the ammonolysis of ethylene oxide. The reaction yields a mixture of monoethanolamine, diethanolamine, and triethanolamine which is separated to obtain the pure products.생산 방법
Diethanolamine is produced with monoethanolamine and triethanolamine by ammonolysis of ethylene oxide; diethanolamine is then separated by distillation (Mullins 1978). In 1984, 166.2 million pounds of diethanolamine were produced in the United States (USTIC 1985).정의
ChEBI: A member of the class of ethanolamines that is ethanolamine having a N-hydroxyethyl substituent.일반 설명
Oily colorless liquid or solid white crystals. Slight rotten fish or ammonia odor. Denser than water.공기와 물의 반응
Water soluble.반응 프로필
2,2'-Iminodiethanol is an aminoalcohol. Amines are chemical bases. They neutralize acids to form salts plus water. These acid-base reactions are exothermic. The amount of heat that is evolved per mole of amine in a neutralization is largely independent of the strength of the amine as a base. Amines may be incompatible with isocyanates, halogenated organics, peroxides, phenols (acidic), epoxides, anhydrides, and acid halides. Flammable gaseous hydrogen is generated by amines in combination with strong reducing agents, such as hydrides. 2,2'-Iminodiethanol is hygroscopic. 2,2'-Iminodiethanol may be sensitive to exposure to air and light. 2,2'-Iminodiethanol can react with oxidizing materials, acids, CO2, copper alloys, aluminum, zinc, galvanized iron and copper.건강위험
The irritant action of diethanolamine on theeyes can be severe. Direct contact of thepure liquid can impair vision. Irritation onthe skin may be mild to moderate. Theacute oral toxicity of this compound waslow in test animals. The toxic symptomsinclude somnolence, excitement, and musclecontraction.LD50 value, oral (mice): 3300 mg/kg
The vapor pressure of diethanolamine isnegligibly low (<0.01 torr at 20°C (68°F)).At ordinary temperature, this compoundshould not cause any inhalation hazard. Themists, fumes, or vapors at high temperatures,however, can produce eye, skin, and respiratory tract irritation.
In contrast to monoethanolamine, dieth anolamine administered to mice at 1125 mg/kg/day caused no change in maternal mortality, litter size, or percentage survival of thepups (Environmental Health Research andTesting 1987).
화재위험
Special Hazards of Combustion Products: Irritating vapors are generated when heated.화학 반응
Reactivity with Water : No reaction; Reactivity with Common Materials: No reaction; Stability During Transport: Stable; Neutralizing Agents for Acids and Caustics: Flush with water; Polymerization: Not pertinent; Inhibitor of Polymerization: Not pertinent.Pharmaceutical Applications
Diethanolamine is primarily used in pharmaceutical formulations as a buffering agent, such as in the preparation of emulsions with fatty acids. In cosmetics and pharmaceuticals it is used as a pH adjuster and dispersant.Diethanolamine has also been used to form the soluble salts of active compounds, such as iodinated organic acids that are used as contrast media. As a stabilizing agent, diethanolamine prevents the discoloration of aqueous formulations containing hexamethylenetetramine-1,3-dichloropropene salts.
Diethanolamine is also used in cosmetics.
색상 색인 번호
Diethanolamine is contained in many products, as a metalworking fuid. Traces may exist in other etha- nolamine-containing fuids.Safety Profile
Poison by intraperitoneal route. Moderately toxic by ingestion and subcutaneous routes. Mildly toxic by skin contact. A severe eye and mild skin irritant. Experimental reproductive effects. Combustible when exposed to heat or flame; can react with oxidizing materials. To fight fire, use alcohol foam, water, Co2, dry chemical. When heated to decomposition it emits toxic fumes such as NOx. See also AMINES.잠재적 노출
Diethanolamine is present in machining and grinding fluids and has been detected in workplace air in the metal manufacturing industry. It was present in bulk cutting fluids at levels ranging from 4 to 5% (Kenyon et al., 1993). Diethanolamine has also been reported to be present in wetting fluids used in road paving. A level of 0.05 mg/m3 was detected in a stationary sample at a slurry machine discharging a bitumen emulsion containing 0.2% of the amine. All personal exposures were below the detection limit (0.02 mg/m3) (Levin et al., 1994). In a German study (1992–94), diethanolamine was measured in samples of metalworking fluids in a range of 0–44% (n = 69). The number of samples with diethanolamine present steadily declined from 90% to 60% over the study period (Pfeiffer et al., 1996).Carcinogenicity
When DEA was administered cutaneously to pregnant rats and rabbits during organogenesis, developmental toxicity (skeletal variations) was observed only in the rat and only at doses causing significant maternal toxicity.The 2003 ACGIH threshold limit valuetime- weighted average (TLV-TWA) is 3ppm (13mg/m3).
신진 대사
Treatment of Wistar or Sherman rats with diethanolamine caused increases in the formation of hepatic phospholipids (Artom et al 1949). In addition, dietary administration led to incorporation of ethanolamine into hepatic phospholipids (Artom et al 1949), and repeated oral administration of diethanolamine in drinking water (one to three wk) at a dose of 320 mg/kg/d was found to reduce the level of incorporation of ethanolamine and choline into hepatic and renal phospholipids in Sprague-Dawley rats (Barbee and H?rtung 1979b).Dermal absorption of diethanolamine is suggested to occur in rats since Nnitrosodiethanolamine was excreted in the urine of male Sprague-Dawley rats which had been administered diethanolamine by dermal application and given nitrite in their drinking water (Preussman et al 1981).
저장
Diethanolamine is hygroscopic and light- and oxygen-sensitive; it should be stored in an airtight container, protected from light, in a cool, dry place.운송 방법
UN2491 Ethanol amine or Ethanolamine solutions, Hazard class: 8; Labels: 8-Corrosive material.Purification Methods
Fractionally distil the amine twice, then fractionally crystallise it from its melt. Its solubility in H2O is 10% at 20o. [Perrin & Dempsey Buffers for pH and Metal Ion Control Chapman & Hall, London 1974, Beilstein 4 H 283, 4 II 729, 4 III 689, 4 IV 1514.]비 호환성
Diethanolamine is a secondary amine that contains two hydroxy groups. It is capable of undergoing reactions typical of secondary amines and alcohols. The amine group usually exhibits the greater activity whenever it is possible for a reaction to take place at either the amine or a hydroxy group.Diethanolamine will react with acids, acid anhydrides, acid chlorides, and esters to form amide derivatives, and with propylene carbonate or other cyclic carbonates to give the corresponding carbonates. As a secondary amine, diethanolamine reacts with aldehydes and ketones to yield aldimines and ketimines. Diethanolamine also reacts with copper to form complex salts. Discoloration and precipitation will take place in the presence of salts of heavy metals.
폐기물 처리
Controlled incineration; incinerator equipped with a scrubber or thermal unit to reduce nitrogen oxides emissionsRegulatory Status
Included in the FDA Inactive Ingredients Database (IV infusions, ophthalmic solutions, and topical preparations). Included in medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients.다이에탄올아민 준비 용품 및 원자재
원자재
준비 용품
softener 101
coconut oil biethyl alcohol acylamine 1︰1 type
fatliquor RCF I/II
4-(PIPERIDINE-1-CARBONYL)PHENYLBORONIC ACID
C.I 형광발색제 220
4-(2-CYANOETHYLAMINOCARBONYL)PHENYLBORONIC ACID
antistatic Agent P
3-(2-CYANOETHYLAMINOCARBONYL)PHENYLBORONIC ACID
1-(3-Chlorophenyl)-4-(3-chloropropyl)piperazine hydrochloride
PIPERAZINE HEXAHYDRATE
MORPHOLINEFATTYACIDSALT
1-(3-Chlorophenyl)piperazine dihydrochloride
N-메틸다이에탄올아민
coconut oil alcohol acylamide
NORMEPERIDINE
N-(2-[4-(4-CHLOROPHENYL)PIPERAZIN-1-YL]ETHYL)-3-METHOXYBENZAMIDE
2-메톡시 에탄올
2-[[4-[[4-[bis(2-hydroxyethyl)amino]-6-chloro-1,3,5-triazin-2-yl]amino]phenyl]azo]-p-cresol
Desulfurizer,high efficiency
사이클렌
dodecylyl diethanol amide 1︰2 type
시클로포스파미드, 모노수화물
4-CYANO-4-PHENYLPIPERIDINE HYDROCHLORIDE
N,N-BIS(2-CHLOROETHYL)-P-TOLUENESULFONAMIDE
2-Bromo-N-(2-bromoethyl)ethanamine
lauroyl bi-ethyl alcohol acylamine 1︰1 type
메클레타민 수화염화물
4-phenyl-1-(p-tolylsulphonyl)piperidine-4-carboxylic acid
Bis(2-chloroethyl)aminophosphonic dichloride
Bis(2-chloroethyl)amine hydrochloride
글리포세이트
dodecay diethanol amine sulfate
이미노 디초산
카세인
케토코나졸
1-(2,6-DIMETHYLPHENYL)PIPERAZINE
Dropropizine
1-(3,5-DICHLOROPHENYL)PIPERAZINE
4-(PYRROLIDIN-1-YLMETHYL)BENZENEBORONIC ACID, PINACOL ESTER 97%1-[4-(4,4,5,5-TETRAMETHYL-1,3,2-DIOXABOROLAN-2-YL)BENZYL]PYRROLIDINE
메클로레타민
다이에탄올아민 공급 업체
글로벌( 868)공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|---|---|---|---|---|
| Shandong Dexiang International Trade Co., Ltd | 15662695772;+8615662691337 |
sales006@dexiangchemical.com | China | 998 | 58 |
| Hebei Mojin Biotechnology Co., Ltd | +8613288715578 |
sales@hbmojin.com | China | 12456 | 58 |
| Henan Fengda Chemical Co., Ltd | +86-371-86557731 +86-13613820652 |
info@fdachem.com | China | 8174 | 58 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 |
info@tianfuchem.com | China | 21691 | 55 |
| Hefei TNJ Chemical Industry Co.,Ltd. | +86-0551-65418679 +86-18949832763 |
info@tnjchem.com | China | 2989 | 55 |
| Tianjin Zhongxin Chemtech Co., Ltd. | +86-022-66880623 +8618622897568 |
sales@tjzxchem.com | China | 559 | 58 |
| Shanxi Naipu Import and Export Co.,Ltd | +86-13734021967 +8613734021967 |
kaia@neputrading.com | China | 1011 | 58 |
| career henan chemical co | +86-0371-86658258 |
sales@coreychem.com | China | 29914 | 58 |
| SHANDONG ZHI SHANG CHEMICAL CO.LTD | +86 18953170293 |
sales@sdzschem.com | China | 2931 | 58 |
| Hubei Jusheng Technology Co.,Ltd. | 18871490254 |
linda@hubeijusheng.com | CHINA | 28180 | 58 |
다이에탄올아민 관련 검색:
에탄올아민 N-메틸다이에탄올아민 트리에탄올아민 에틸렌디아민테트라아세트산, 이나트륨 염 트랜스-1,2-사이클로헥산디아민테트라아세트산 1,6-디아미노헥산-N,N,N′,N′-테트라아세틱 산 1,1'-이미노비스-2-프로판올 히드록시에틸렌디아민트리아세트 산 비스-트리스 부틸말론산디에틸에스테르 3,6,9,12-테트라키스(카복시메틸)-3,6,9,12-테트라아자테트라데케인다이오산 알리자린 플루오린 블루 나이트릴로트라이아세트산 다이에탄올아민 2-2'-(페닐이미노)디에탄올 티몰프탈레인컴플렉손 디에틸렌트리아민펜타아세트산 N-(트리스(하이드록시메틸)메틸)글리신