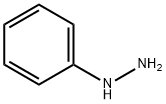
페닐히드라진
|
|
페닐히드라진 속성
- 녹는점
- 18-21 °C (lit.)
- 끓는 점
- 238-241 °C (lit.)
- 밀도
- 1.098 g/mL at 25 °C (lit.)
- 증기 밀도
- 4.3 (vs air)
- 증기압
- <0.1 mm Hg ( 20 °C)
- 굴절률
- n
20/D 1.607(lit.)
- 인화점
- 192 °F
- 저장 조건
- Store below +30°C.
- 용해도
- 묽은 산에 용해됩니다.
- 물리적 상태
- 가루
- 산도 계수 (pKa)
- 8.79(at 15℃)
- 색상
- 흰색에서 약간 파란색 또는 연한 베이지색
- 폭발한계
- 1.1%(V)
- 수용성
- 145g/L(20℃)
- 감도
- Air & Light Sensitive
- Merck
- 14,7293
- BRN
- 606080
- 노출 한도
- TLV-TWA skin 0.1 ppm (0.44 mg/m3) (ACGIH), 5 ppm (22 mg/m3) (OSHA); STEL 10 ppm (44 mg/m3) (OSHA); carcinogenicity: A2-Suspected Human Carcinogen (ACGIH), Carcinogen (NIOSH). .
- Dielectric constant
- 7.2(23℃)
- 안정성
- 안정적이지만 햇빛에 분해될 수 있습니다. 공기나 빛에 민감할 수 있습니다. 강한 산화제, 금속 산화물과 호환되지 않습니다.
- CAS 데이터베이스
- 100-63-0(CAS DataBase Reference)
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| 위험품 표기 | T,N | ||
|---|---|---|---|
| 위험 카페고리 넘버 | 45-23/24/25-36/38-43-48/23/24/25-50-68 | ||
| 안전지침서 | 53-45-61 | ||
| 유엔번호(UN No.) | UN 2572 6.1/PG 2 | ||
| WGK 독일 | 3 | ||
| RTECS 번호 | MV8925000 | ||
| F 고인화성물질 | 8-10-23 | ||
| 자연 발화 온도 | 345 °F | ||
| TSCA | Yes | ||
| HS 번호 | 2928 00 90 | ||
| 위험 등급 | 6.1 | ||
| 포장분류 | II | ||
| 유해 물질 데이터 | 100-63-0(Hazardous Substances Data) | ||
| 독성 | LD50 orally in Rabbit: 188 mg/kg | ||
| IDLA | 15 ppm | ||
| 기존화학 물질 | KE-28379 | ||
| 유해화학물질 필터링 | 97-1-335 | ||
| 중점관리물질 필터링 | 별표1-49 | ||
| 암, 돌연변이성물질 필터링 | 35 | ||
| 함량 및 규제정보 | 물질구분: 유독물질; 혼합물(제품)함량정보: 페닐히드라진 및 이를 1% 이상 함유한 혼합물 |
페닐히드라진 C화학적 특성, 용도, 생산
용도
보통 염산염으로 사용되는 경우가 많다. 안티피린의 원료이고 알데하이드·케톤·당류등에 대한 시약으로 중요하며, 또 인의 정량시약으로도 사용된다. 다양한 염료와 의약품의 합성 과정에서 중간물질로 얻어지는 인돌을 만드는 데 사용된다.용도
1) 알데히드 및 케톤과 반응해서 페닐히드라존의 결정을 생성하고, 또 각종의 종류 또는 α-케톨과 반응해서 오사존의 결정을 생성하므로, 이들 화합물의 검출 확인에 널리 이용된다. 2) 또 피라졸, 피리다진 등의 헤테로 고리를 갖는 화합물의 합성 원료로서 사용된다. 3) 인의 정량 시약으로서 사용된다. 인을 몰리브덴산암모늄으로 하고, 이것을 이 시약으로 환원해서 얻어지는 몰리브덴 블루의 청색을 인의 비색 분석에 이용한다.화학적 성질
Phenylhydrazine is a colorless to pale yellow liquid or solid with a weak aromatic odor. It is soluble in water (values ranging from 145 to 837 g/litre at 24 °C have been reported) and is miscible with alcohol, ether, chloroform, benzene, and acetone. The conversion factor for phenylhydrazine is 1 ppm = 4.5 mg/m3 (at 20 °C, 101 kPa).물리적 성질
Yellow monoclinic crystals with a faint, aromatic odor. Turns red-brown on exposure to air.용도
Phenylhydrazine is used in the manufactureof dyes and pharmaceuticals; as a stabilizerfor explosive; as a reagent for aldehydes,ketones, and sugars in chemical analysis; andin organic synthesis.정의
ChEBI: A phenylhydrazine that is the monophenyl derivative of hydrazine.일반 설명
Pale yellow crystals. Melting point 66°F. Becomes an oily liquid. Toxic by ingestion, inhalation and skin absorption. Flash point 192°F. Autoignition temperature 345°F. Soluble in alcohol.공기와 물의 반응
When exposed to air becomes red-brown. Slightly denser than water and slightly soluble in water.반응 프로필
Phenylhydrazine may ignite spontaneously when in contact with oxidants such as hydrogen peroxide or nitric acid, oxides of iron or copper, or manganese, lead, copper or their alloys. Will not polymerize [USCG, 1999].건강위험
Phenylhydrazine is a moderate to highlytoxic compound and a carcinogen. Acutetoxic symptoms include hematuria, changesin liver and kidney, vomiting, convulsions,and respiratory arrest. Additional symptomsare lowering of body temperature and fallin blood pressure. Chronic exposure to thiscompound caused hemolytic anemia andsignificant body weight loss in rats. Anoral administration of 175 mg/kg was lethalto mice. An oral LD50 value in rat is188 mg/kgPhenylhydrazine caused adverse repro ductive effect when dosed intraperitoneallyin pregnant mice. It caused jaundice, anemia,and behavioral deficit in offspring
Oral or subcutaneous administration ofphenylhydrazine or its hydrochloride produced lung and liver tumors in experimental animals. ACGIH lists this compoundas a suspected human carcinogen. The evidence of carcinogenicity of this compoundin human is inadequate.
화재위험
Combustible liquid; flash point (closed cup) 89°C (192°F). It reacts with lead dioxide and strong oxidizing compounds vigorous to violently.Safety Profile
Confirmed carcinogen with experimental carcinogenic data. Poison by ingestion, subcutaneous, and intravenous routes experimental reproductive effects Mutation data reported. Ingestion or subcutaneous injection can cause hemolysis of red blood cells. Other effects are damage to the spleen, liver, kidneys, and bone marrow. The most common effect of occupational exposure is the development of dermatitis, which, in sensitized persons, may be quite severe. Systemic effects include anemia and general weakness, gastrointestinal dsturbances, and injury to the kidneys. Flammable when exposed to heat, flame, or oxidizers. To fight fire, use alcohol foam. Violent reaction with 2phenylamino-3-phenyloxazirane. Reacts with perchloryl fluoride to form an explosive product. Vigorous reaction with lead(Ⅳ) oxide. Used as a chemical reagent, in organic synthesis, and in the manufacture of dyes and drugs. Dangerous; when heated to decomposition it emits highly toxic fumes of NOx; can react with oxidizing materials잠재적 노출
Phenylhydrazine is a widely used reagent in conjunction with sugars, aldehydes, and ketones. In addition, to its use in the synthesis of dyes; pharmaceuticals, such as antipyrin; cryogenin, and pyramidone; and other organic chemicals. The hydrochloride salt is used in the treatment of polycythemia vera.Carcinogenicity
Mice given 1mg phenylhydrazine orally each day, 7 days/week for 42 weeks had an increased incidence of total lung tumors. The incidence of malignant tumors also increased. In another study, mice administered 0.01% of the hydrochloride salt in drinking water (average of 0.63–0.81 mg/day) over their lifetimes had an increased incidence of blood vessel tumors. In contrast, mice given phenylhydrazine orally 5 days/week for 40 weeks at doses of 0.5 mg during the first 5 weeks and 0.25 mg thereafter failed to develop tumors.Higher doses could not be tested because of the marked anemia encountered. No leukemias occurred in mice treated with phenylhydrazine, and pulmonary tumors were infrequent . Oral administration or intraperitoneal injection (eight doses, each of 2.9 mg) of phenylhydrazine hydrochloride in mice failed to increase either pulmonary tumors or leukemia. The carcinogenic activity of hydrazine itself has been closely linked to its toxicity, that is, its irritant effect. The mechanism of action, it is postulated, is indirect alkylation of DNA, which is also closely connected to the toxic end point.운송 방법
UN2572 Phenylhydrazine, Hazard Class: 6.1; Labels: 6.1-Poisonous materials.Purification Methods
Purify phenylhydrazine by chromatography, then crystallise it from pet ether (b 60-80o)/*benzene. Store it in the dark under N2 as it turns yellow, then red, on exposure to air. It is best stored as the hydrochloride salt; see below. [Coleman Org Synth Coll Vol I 442 1941, Shaw & Stratton J Chem Soc 5004 1962, Beilstein 15 IV 50.]비 호환성
Phenylhydrazine is very reactive with carbonyl compounds, strong oxidizers; strong bases; alkali metals; ammonia, lead dioxide (violent). Attacks copper salts, nickel, and chromates.폐기물 처리
Controlled incineration whereby oxides of nitrogen are removed from the effluent gas by scrubber, catalytic or thermal device.페닐히드라진 준비 용품 및 원자재
원자재
준비 용품
BISPYRAZOLONE
2-(3-CHLORO-4-FLUOROPHENYL)INDOLE
5-METHYL-1-PHENYL-1H-PYRAZOLE-4-CARBOXYLIC ACID
클로펜테진
2-(4-FLUOROPHENYL)-1H-INDOLE-3-CARBALDEHYDE
2-Phenyl-1H-indole-1-acetic acid ,97%
1,2,3,4-Tetrahydro-9-methylcarbazol-4-one
3,5-DIMETHYL-1-PHENYL-1H-PYRAZOLE-4-CARBALDEHYDE
1-PHENYL-1H-PYRAZOLE-4-CARBALDEHYDE
SULFAPHENAZOLE
1-페닐-3-피라졸리돈
ETHYL METHANESULFONYLACETATE
ETHYL 5-METHYL-1-PHENYL-1H-PYRAZOLE-4-CARBOXYLATE
5-AMINO-1-PHENYLPYRAZOLE
5-(TRIFLUOROMETHYL)-1-PHENYL-1H-PYRAZOLE-4-CARBALDEHYDE
Triazophos E.C.
1-Methyl-2-phenylindole
4-(1H-INDOL-2-YL)-PHENYLAMINE
4-BROMO-5-(TRIFLUOROMETHYL)-1-PHENYL-1H-PYRAZOLE
(5-METHYL-1-PHENYL-1H-PYRAZOL-4-YL)METHANOL
6-(4-FLUOROPHENYL)INDOLE
1-Methyl-2-phenylindole-3-carboxaldehyde
3-METHYL-1-PHENYLPYRAZOLE
피라진아마이드
2-(4-ETHOXY-PHENYL)-1H-INDOLE
2-(4-BROMO-PHENYL)-1H-INDOLE
ethyl 2-(2-phenyl-1H-indol-1-yl)acetate
1-PHENYLPYRAZOLE
5-(TRIFLUOROMETHYL)-1-PHENYL-1H-PYRAZOLE
2-(4-CHLOROPHENYL)INDOLE
1,5-디페닐카보노히드라지드
5-AMINO-4-CARBETHOXY-1-PHENYLPYRAZOLE
5-CHLORO-1,3-DIPHENYL-1H-PYRAZOLE-4-CARBALDEHYDE
1,2,3,9-Tetrahydro-4(H)-carbazol-4-one
2-페닐-2-이미다졸린
2-(2-NAPHTHYL)-1H-INDOLE-3-CARBALDEHYDE
2,2,2-TRIFLUORO-1-(3-(TRIFLUOROMETHYL)-1-PHENYL-1H-PYRAZOL-4-YL)ETHANONE
염화 2,3,5-트리페닐 테트라졸륨
2-Phenylindole
5-AMINO-1-PHENYLPYRAZOLE-4-CARBONITRILE
페닐히드라진 공급 업체
글로벌( 402)공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|---|---|---|---|---|
| Hebei Mojin Biotechnology Co., Ltd | +8613288715578 |
sales@hbmojin.com | China | 12456 | 58 |
| Capot Chemical Co.,Ltd. | 571-85586718 +8613336195806 |
sales@capotchem.com | China | 29797 | 60 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 |
info@tianfuchem.com | China | 21691 | 55 |
| Hangzhou FandaChem Co.,Ltd. | 008657128800458; +8615858145714 |
fandachem@gmail.com | China | 9348 | 55 |
| Hefei TNJ Chemical Industry Co.,Ltd. | +86-0551-65418679 +86-18949832763 |
info@tnjchem.com | China | 2989 | 55 |
| Shanxi Naipu Import and Export Co.,Ltd | +86-13734021967 +8613734021967 |
kaia@neputrading.com | China | 1011 | 58 |
| career henan chemical co | +86-0371-86658258 |
sales@coreychem.com | China | 29914 | 58 |
| Hubei Jusheng Technology Co.,Ltd. | 18871490254 |
linda@hubeijusheng.com | CHINA | 28180 | 58 |
| Hebei Guanlang Biotechnology Co., Ltd. | +86-19930503282 |
alice@crovellbio.com | China | 8823 | 58 |
| Hubei xin bonus chemical co. LTD | 86-13657291602 |
linda@hubeijusheng.com | CHINA | 22968 | 58 |