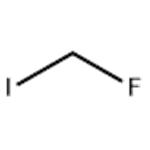
- fluoro-iodo-methane
-
- $101.00 / 1KG
-
2023-12-29
- CAS:373-53-5
- Min. Order: 1KG
- Purity: 99%
- Supply Ability: g-kg-tons, free sample is available
Related articles - Summary of fluoroiodomethane uses
- Fluoroiodomethane (CH2FI) has emerged as an easy-to-handle, non-ozone-depleting agent and readily available platform for monof....
- Jan 9,2024
|
| | Fluoroiodomethane Basic information |
| Product Name: | Fluoroiodomethane | | Synonyms: | fluoro-iodo-methane;Fluoroiodomethane 95%;Methane, fluoroiodo- (6CI,8CI,9CI);Fluoroiodomethane (ca. 2mol/L in Acetonitrile);Fluoromethyl iodide;Fluor(iod)methan | | CAS: | 373-53-5 | | MF: | CH2FI | | MW: | 159.93 | | EINECS: | | | Product Categories: | | | Mol File: | 373-53-5.mol | 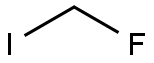 |
| | Fluoroiodomethane Chemical Properties |
| Boiling point | 53.4℃ | | density | 2.366 g/cm3 (20 ºC) | | refractive index | 1.491 (589.3 nm 20℃) | | form | clear liquid | | color | Colorless to Light yellow | | Stability: | Volatile | | InChI | InChI=1S/CH2FI/c2-1-3/h1H2 | | InChIKey | XGVXNTVBGYLJIR-UHFFFAOYSA-N | | SMILES | C(F)I | | CAS DataBase Reference | 373-53-5 |
| | Fluoroiodomethane Usage And Synthesis |
| Uses | Fluoroiodomethane is an synthetic precursor for the fluoromethylation of radiopharmaceuticals. | | Properties and Applications | Fluoroiodomethane is a commercially available liquid with a boiling point of 53.4 °C. This reagent is easy to handle and represents a non-ozone-depleting alternative to the frequently used CFC (chlorofluorocarbons) and HBFC (hydrobromofluorocarbons). This reagent is featured by two carbon–halogen bonds (C–I and C–F) characterized by diverse reactivity. The bond dissociation energy (BDE) of C–I (233 kJ mol?1) compared to that of the C–F bond (460 kJ mol?1) enables a selective activation of CH2FI for multiple pathways delivering the fluoromethyl fragment unit. Indeed, the C–I bond can be engaged in lithium exchange protocols, transition-metal catalyzed reactions, nucleophilic substitutions, and radical pathways[1].
| | References | [1] Marco Colella . “The synthetic versatility of fluoroiodomethane: recent applications as monofluoromethylation platform.” Organic Biomolecular Chemistry 20 23 (2022): Pages 4669-4680. |
| | Fluoroiodomethane Preparation Products And Raw materials |
|