- Tucatinib
-

- $0.00 / 1kg
-
2023-12-21
- CAS:937263-43-9
- Min. Order: 1kg
- Purity: 99%,single impurity<0.1
- Supply Ability: 1 ton
- Tucatinib
-
- $0.00 / 1kg
-
2023-12-01
- CAS:937263-43-9
- Min. Order: 1kg
- Purity: >99.5% by HPLC
- Supply Ability: 100kg/month
- Irbinitinib
-
- $0.00 / 1KG
-
2023-06-19
- CAS:937263-43-9
- Min. Order: 1KG
- Purity: 98%
- Supply Ability: 1000KGS
Related articles - Synthesis of Tucatinib
- Tucatinib was synthesised in a two-step reaction by an aniline fragment. Tucatinib is an orally bioavailable tyrosine kinase i....
- Jan 8,2024
|
| | Tucatinib Basic information |
| Product Name: | Tucatinib | | Synonyms: | ONT-380);N4-(4-([1;N6-(4,5-Dihydro-4,4-dimethyl-2-oxazolyl)-N4-[3-methyl-4-([1,2,4]triazolo[1,5-a]pyridin-7-yloxy)phenyl]-4,6-quinazolinediamine;Irbinitinib;Irbinitinib(ARRY380);(R)-N6-(4-Methyl-4;5-dihydrooxazol-2-yl)-N4-(3-Methyl-4-(thiazol-2-ylMethoxy)phenyl)quinazoline-4;6-diaMine | | CAS: | 937263-43-9 | | MF: | C26H24N8O2 | | MW: | 480.52 | | EINECS: | 200-001-8 | | Product Categories: | Inhibitors;API | | Mol File: | 937263-43-9.mol | 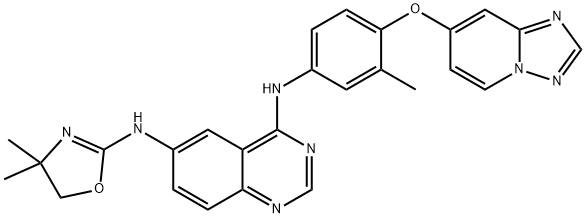 |
| | Tucatinib Chemical Properties |
| density | 1.41±0.1 g/cm3(Predicted) | | storage temp. | Store at -20°C | | solubility | DMF:1.0(Max Conc. mg/mL);2.08(Max Conc. mM)
DMSO:49.0(Max Conc. mg/mL);101.97(Max Conc. mM)
DMSO:PBS (pH 7.2) (1:2):0.33(Max Conc. mg/mL);0.69(Max Conc. mM) | | form | A crystalline solid | | pka | 6.61±0.70(Predicted) | | InChIKey | SDEAXTCZPQIFQM-UHFFFAOYSA-N | | SMILES | N1=C2C(C=C(NC3=NC(C)(C)CO3)C=C2)=C(NC2=CC=C(OC3C=CN4N=CN=C4C=3)C(C)=C2)N=C1 |
| | Tucatinib Usage And Synthesis |
| Description | Tucatinib is also known as ARRY-380 or Irbinitinib. ARRY-380 is an orally inhibitor of human epidermal growth factor receptor tyrosine kinase ErbB-2 (also called HER2) with potential antineoplastic activity. It selectively binds to and inhibits the phosphorylation of ErbB-2, which may prevent the activation of ErbB-2 signal transduction pathways, resulting in growth inhibition and death of ErbB-2-expressing tumor cells. ErbB-2 is overexpressed in a variety of cancers and plays an important role in cellular proliferation and differentiation. Therefore, ARRY-380 may be an alternative treatmentmethod for the treatment of HER2+ cancers. | | Characteristics | Year of discovery: 2006
Year of introduction: 2020
Discovered by: Array BioPharma
Developed by: Array BioPharma
Primary targets: HER2
Binding type: I/II
Class: receptor tyrosine kinase
Treatment: HER2-positive breast cancer
Other names: ONT-380, ARRY-380
Oral bioavailability = not reported
Elimination half-life = 5.4 hours
Protein binding = 97% | | Uses | Tucatinib, a highly selective HER2 inhibitor, was approved in 2020 as a combination therapy with trastuzumab and capecitabine for the treatment of patients with advanced unresectable or metastatic HER2-positive breast cancer, including patients with brain metastases, who have received one or more prior anti-HER2-based regimens in the metastatic setting. | | storage | Store at -20°C | | Mode of action | Tucatinib binds to tyrosine kinase (an enzyme) of HER2, reducing PI3-kinase and MAP-kinase signaling. In vitro, tucatinib inhibits phosphorylation of HER2 and HER3, resulting in inhibition of downstream MAPK and AKT signaling and cell proliferation, and showed anti-tumor activity in HER2 expressing tumor cells. In vivo, tucatinib inhibited the growth of HER2 expressing tumors. The combination of tucatinib and trastuzumab showed increased anti-tumor activity in vitro and in vivo compared to either drug alone. | | References | [1] PATRICE A LEE. In VivoActivity of ARRY-380, a Potent, Small Molecule Inhibitor of ErbB2 in Combination with Trastuzumab, Docetaxel or Bevacizumab.[J]. Cancer research, 2009, 69 1: 5104-5104. DOI:10.1158/0008-5472.SABCS-09-5104.
[2] S. MOULDER. Abstract A143: ARRY-380, a selective HER2 inhibitor: From drug design to clinical evaluation.[J]. Molecular Cancer Therapeutics, 2011, 10 1. DOI:10.1158/1535-7163.TARG-11-A143.
[3] V. DINKEL. Abstract 852: ARRY-380, a potent, small molecule inhibitor of ErbB2, increases survival in intracranial ErbB2+ xenograft models in mice[J]. Cancer research, 2012, 72 1: 852-852. DOI:10.1158/1538-7445.AM2012-852.
[4] KOCIE?SKI P. Synthesis of Tucatinib[J]. Synfacts, 2019, 24 1: 0965. DOI:10.1055/s-0039-1690496.
[5] RASHMI K MURTHY. Tucatinib, Trastuzumab, and Capecitabine for HER2-Positive Metastatic Breast Cancer.[J]. New England Journal of Medicine, 2020, 382 7: 597-609. DOI:10.1056/NEJMoa1914609.
[6] RASHMI MURTHY. Tucatinib with capecitabine and trastuzumab in advanced HER2-positive metastatic breast cancer with and without brain metastases: a non-randomised, open-label, phase 1b study.[J]. Lancet Oncology, 2018: 880-888. DOI:10.1016/S1470-2045(18)30256-0. |
| | Tucatinib Preparation Products And Raw materials |
|