|
|
| | TUNGSTEN(VI) OXYCHLORIDE Basic information |
| Product Name: | TUNGSTEN(VI) OXYCHLORIDE | | Synonyms: | TUNGSTEN OXYCHLORIDE;TUNGSTEN(VI) OXYCHLORIDE;tungsten(vi) chloride oxide;Tungsten(VI) chloride oxide, Tungsten(VI) oxide chloride;tungsten tetrachloride oxide;TUNGSTEN OXIDE CHLORIDE;TUNGSTEN OXYCHLORIDE 99%;Tungsten chloride oxide(WCl4O) | | CAS: | 13520-78-0 | | MF: | Cl4OW | | MW: | 341.65 | | EINECS: | 236-863-7 | | Product Categories: | | | Mol File: | 13520-78-0.mol | 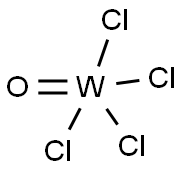 |
| | TUNGSTEN(VI) OXYCHLORIDE Chemical Properties |
| Melting point | 211 °C(lit.) | | Boiling point | 337°C | | density | 11.920 | | solubility | reacts with H2O; soluble in benzene, CS 2 | | form | red hygroscopic crystals | | color | red hygroscopic crystals, crystalline | | Water Solubility | decomposed by H2O; soluble CS2, benzene [KIR83] [HAW93] | | Sensitive | air sensitive, moisture sensitive |
| | TUNGSTEN(VI) OXYCHLORIDE Usage And Synthesis |
| Description | Tungsten(VI) oxytetrachloride, WOCl4, is a diamagnetic solid used to prepare other tungsten complexes. The orange-colored compound is soluble in nonpolar solvents, while it reacts with alcohols and water and forms adducts with Lewis bases. | | Chemical Properties | Dark-red, acicular crystals. Decomposed
by water and moist air. Keep in sealed glass
container. Bp 227.5C, mp 211C, d 11.92. Soluble
in carbon disulfide. | | Uses | Incandescent lamps. | | Preparation | It consists of weakly associated square pyramidal monomers.
WOCl4 is formed from WO3 or WCl6.
WO3 + 2SOCl2→WOCl4+2SO2
WCl6 + (Me3Si )2O→WOCl4+ 2Me3SiCl | | Hazard | Irritant. |
| | TUNGSTEN(VI) OXYCHLORIDE Preparation Products And Raw materials |
|