- Ticagrelor
-

- $0.00 / 25Kg/Bag
-
2024-09-19
- CAS:274693-27-5
- Min. Order: 2Kg/Bag
- Purity: 99% up, High Density
- Supply Ability: 20 tons
- TICAGRELOR
-

- $100.00 / 1kg
-
2024-09-19
- CAS:274693-27-5
- Min. Order: 10kg
- Purity: 99%
- Supply Ability: 10000kg
- Ticagrelor
-

- $0.00 / 1Kg/Bag
-
2024-09-18
- CAS:274693-27-5
- Min. Order: 1KG
- Purity: 98%-102%
- Supply Ability: 100kgs
Question and answer - Q:Is ticagrelor same as Plavix ?
- A:Brilinta (ticagrelor) and Plavix (clopidogrel) are used to prevent blood clots in patients with heart issues and reduce the ri....
- Jun 7,2024
|
| Product Name: | TICAGRELOR | | Synonyms: | 1,2-Cyclopentanediol, 3-[7-[[(1R,2S)-2-(3,4-difluorophenyl)cyclopropyl]amino]-5-(propylthio)-3H-1,2,3-triazolo[4,5-d]pyrimidin-3-yl]-5-(2-hydroxyethoxy)-, (1S,2S,3R,5S)-;Ticagrelor, >=98%;Ticagrelo;Ticarelor;Brilique;(1S,2S,3R,5S)-3-[7-[[(1R,2S)-2-(3,4-difluorophenyl)cyclopropyl]amino]-5-propylsulfanyltriazolo[4,5-d]pyrimidin-3-yl]-5-(2-hydroxyethoxy)cyclopentane-1,2-diol;TICAGRELOR;(1S,2S,3R,5S)-3-[7-[[(1R,2S)-2-(3,4-Difluorophenyl)cyclopropyl]amino]-5-(propylthio)-3H-1,2,3-triazolo[4,5-d]pyrimidin-3-yl]-5-(2-hydroxyethoxy)-1,2-cyclopentanediol | | CAS: | 274693-27-5 | | MF: | C23H28F2N6O4S | | MW: | 522.57 | | EINECS: | 619-540-9 | | Product Categories: | AZD6140;Ticagrelor;Cardiovascular APIs;Aromatics;Heterocycles;Intermediates & Fine Chemicals;Pharmaceuticals;Sulfur & Selenium Compounds;API;274693-27-5 | | Mol File: | 274693-27-5.mol | 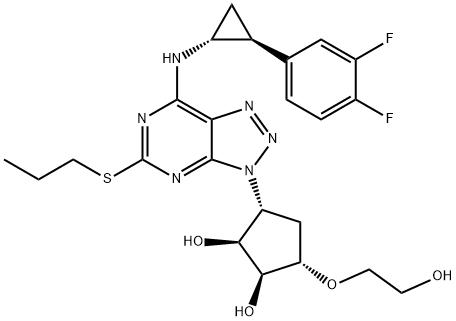 |
| | TICAGRELOR Chemical Properties |
| Melting point | 138-140°C | | Boiling point | 777.6±70.0 °C(Predicted) | | density | 1.67 | | storage temp. | Keep in dark place,Inert atmosphere,Store in freezer, under -20°C | | solubility | Methanol (Slightly) | | pka | 13.26±0.70(Predicted) | | form | Solid | | color | White to Off-White | | Stability: | Hygroscopic | | InChIKey | OEKWJQXRCDYSHL-FNOIDJSQSA-N | | SMILES | [C@@H]1(O)[C@@H](OCCO)C[C@@H](N2C3C(N=N2)=C(N[C@@H]2C[C@H]2C2=CC=C(F)C(F)=C2)N=C(SCCC)N=3)[C@@H]1O |
| | TICAGRELOR Usage And Synthesis |
| Indications and Usage | Ticagrelor, a new platelet aggregation inhibitor successfully developed by AstraZeneca (U.S.), is the world’s first reversible combination orally-administered P2Y12 adenosine diphosphate receptor antagonist.
It is used to reduce cardiovascular death and heart attacks in patients with acute coronary syndrome (ACS.) Rapid onset after oral administration, and can effectively improve symptoms of patients with ACS. Thienopyridines is a reversible P2Y12 inhibitor, so it is particularly applicable towards patients who need to undergo anticoagulant therapy before surgery.
| | Mechanisms of Action | Reversibly acts on the 2 purine receptor (purinoceptor 2, P2,) subtype P2Y12, of vascular smooth muscle cells (VSMC,) does not require metabolic activation, and has a significant inhibitory effect on platelet aggregation induced by adenosine diphosphate (ADP.)
| | Clinical Research | Ticagrelor was approved by the U.S. Food and Drug Administration (FDA) on the basis of PLATO clinical studies. PLATO was a 43 country, 862 center randomized, double-blind, multi-centered study which included 18,624 patients hospitalized for ACS. Patients were randomly given either aspirin plus ticagrelor, or aspirin plus clopidogrel, two antiplatelet regimens. The study showed a 16% reduction in risk of heart attack, stroke, or death in the group taking Ticagrelor compared to those who took clopidogrel. The PLATO study demonstrated the benefits of Ticagrelor for patient survival, and is currently widely recommended as a treatment strategy for ACS. In addition, at the same time it reduces operative myocardial infarction and thrombosis after stent implantation, it also does not increase the overall risk of fatal bleeding and other serious adverse effects.
Ticagrelor however has two shortcomings. First, it must be taken twice a day, compared to once a day for clopidogrel, and secondly, it may cause adverse effects such as breathing difficulties, significantly more than with the latter. Its half-life is only 12 hours, and taken twice a day, a challenge for patients with poor compliance. In practice, it has also been found that around 20% of patients treated with clopidogrel do not follow their prescriptions, and these patients are even less likely to take the prescribed doses when using ticagrelor. Thus, as a rapidly reversible drug, direct withdrawal will likely increase the risk of acute thrombosis, causing myocardial infection or stroke.
| | Description | Ticagrelor is a reversible antagonist of the platelet purinergic P2Y12 receptor (Ki = 14 nM; IC50 = 1.8 μM), which is the main receptor responsible for ADP-induced platelet aggregation. It functions by directly changing the conformation of the P2Y12 receptor to inhibit ADP binding. Formulations containing ticagrelor have been used to reduce the rate of thrombotic cardiovascular events in patients with acute coronary syndrome. | | Description | In December 2010, the P2Y12 receptor antagonist ticagrelor (also
known as AZD6140) was approved in Europe for the treatment of acute
coronary syndrome (ACS), a condition that covers several clinical symptoms
with the potential to cause acute myocardial ischemia (MI).
ADP binds to two purinergic receptors, the P2Y1 and P2Y12 receptors. The
action of ADP binding to the P2Y12 receptor results in activation of the GP
Ⅱb/Ⅲa (integrin) receptor.GP Ⅱb/Ⅲa initiates and prolongs platelet aggregation,
which in turn results in the cross-linking of platelets through fibrin
and finally thrombus formation. Inhibition of ADP stimulation of the P2Y12
receptor has been found to be an effective strategy for managing the atherothrombotic
events associated with ACS and potentially resulting from
percutaneous coronary intervention (PCI, stent implantation) . | | Chemical Properties | White Solid | | Originator | Astra-Zeneca (United Kingdom) | | Uses | Ticagrelor, the first reversible oral P2Y12 receptor antagonist, provides faster, greater, and more consistent ADP-receptor inhibition than Clopidogrel. Used in the treatment of acute coronary syndromes (ACS). | | Uses | Ticagrelor is the first reversibly binding oral P2Y12 receptor antagonist, also inhibits CYP2C9 and 4-hydroxylation with IC50 of 10.5 μM and 8.2 μM respectively | | Definition | ChEBI: A triazolopyrimidine that is an adenosine isostere; the cyclopentane ring is similar to ribose and the nitrogen-rich [1,2,3]triazolo[4,5-d]pyrimidine moiety resembles the nucleobase adenine. A platelet aggregation inhibitor which is used for p
evention of thromboembolic events in patients with acute coronary syndrome. | | Brand name | Brilique and Possia in the European Union | | Clinical Use | Ticagrelor, discovered and developed by AstraZeneca, is a platelet
adenosine diphosphate (ADP) P2Y12 (P2T) reversible receptor
antagonist approved in the E.U. in 2010 and launched in Germany
and the UK in 2011 for the treatment of patients with acute coronary
syndromes (ACS). It was approved in the U.S. and Canada in
2011 following successful clinical trial results in patients with
ACS which showed it to be superior to preexisting drugs for reducing death due to vascular causes. Ticagrelor is an oral drug
indicated for use in combination with acetylsalicylic acid (aspirin)
for the prevention of atherothrombotic events in adult patients
with ACS (unstable angina, non-ST elevation myocardial infarction
(NSTEMI), or ST elevation myocardial infarction (STEMI)). Unlike
its competitors prasugrel and clopidogrel, which require bioactivation,
ticagrelor is not a prodrug and does not require in vivo activation.
It has a rapid onset of action, relatively rapid reversibility,
greater potency, and exhibits consistency in platelet inhibition.
Following dosing, ticagrelor reaches Cmax in about 1.5 h, with formation
of a major metabolite with equipotent intrinsic activity to
the parent compound. | | Synthesis | The initial discovery of the drug
and SAR studies were published in 2007, including the initial discovery
patent applications. Since then, a number of patents
have been published with various improvements made for largescale
synthesis of the drug. While the molecule has been
synthesized using various modifications of the common intermediates,
the large-scale preparation proceeds via a convergent strategy
involving the coupling of three key intermediates as shown in the Scheme below.
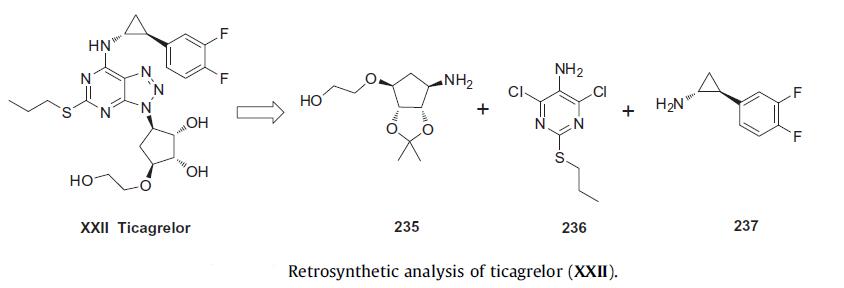
Several routes to the synthesis of cyclopentyl amino alcohol 235
have been reported. Most of these routes are based on reaction of
cyclopentene acetate 238 with the appropriate amine, which is
commercially available. Interestingly, one route targeting
deuterated ticagrelor used a nitroxide Diels¨CAlder reaction with
cyclopentadiene to incorporate the amine into the ring system.
The most likely process-scale preparation of the key cyclopentyl
amine required for ticagrelor is highlighted in the scheme below.

Commercially available enantiopure acetate 238 was reacted
with sodium di-tert-butyloxy diimide under catalytic palladiummediated
amination conditions to give bis-Boc amide 239 in 92%
yield. Dihydroxylation of cyclopentene 239 using catalytic osmium
tetraoxide and N-methyl morpholine N-oxide (NMO) in THF/water
quantatively resulted in the cis-diol 240. The free amine was liberated
with 6 N HCl followed by in situ ketalizaion of the cis-diol
hydrochloride salt 241 in 92% yield. Cbz carbamate 242 was quantitatively
synthesized from 241 under standard conditions. Alcohol
242 was treated with potassium t-butoxide and bromoethyl acetate
(243), the ester intermediate of which was reduced in situ
with lithium borohydride to alcohol 244 in 86% overall yield (two steps). Hydrogenolysis at 1.2 bar of hydrogen pressure with
5% Pd/C gave amino alcohol intermediate 235 in 83% yield. This
amine (235) was mixed with oxalic acid to provide the oxalate salt
in 82% yield, which was subsequently used for the final synthesis of
ticagrelor.
The large-scale preparation of ticagrelor necessitated the synthesis
of dichloroamino pyrimidine thioether 236, for which there
are several reported routes. The synthesis is initiated with
the construction of thiol barbituric acid 247 (Scheme below). This
intermediate was formed from the reaction of dimethyl malonate
(245) with thiourea (246) in the presence of sodium methoxide.
These conditions provided the sodium salt of the pyrimidone thiol
247 in 83% yield, which was isolated via filtration from the reaction
mixture. Salt 247 was then reacted with propyliodide in aqueous
methanolic sodium hydroxide followed by HCl quench to provide
the desired thioether 248 in 76% yield. Nitration of pyrimidinol thioether
248 was achieved by treatment with fuming nitric acid in
acetic acid, furnishing the nitro pyrimidinol 249 in 75% yield. Subsequent
bis-chlorination with POCl3 converted 249 to dichloropyrimidine
thioether 250 in near quantitative yield. In an earlier
publication, a selective reduction of the nitro dichloropyrimide thioether
250 was demonstrated by hydrogenation at 3 bar hydrogen
pressure using 3%Pt/0.6%V/C catalyst to provide the amino dichloropyrimidine
thioether 236 in �95% yield. It is also of note that
for the larger kilo-scale reaction, selective hydrogenation was
accomplished with Pt/V/C (2% Pt; 1% V on carbon) catalyst with
8 bar of hydrogen pressure to give the crude amino dichloropyrimidine
thioether 236.

While a number of routes have been described for the preparation
of cyclopropyl amine intermediate 237,184¨C187,193¨C196 the large
scale route used is described (Scheme below).195 Condensation of
malonic acid and 3,4-difluorobenzaldehyde (251) with piperidine
in pyridine gave acid 252 in 88% yield after acidic work-up. Acid
chloride 253 was prepared using thionyl chloride, which was
followed by esterification with L-menthol and pyridine to give Lmenthol
ester 254 in 93% over 2 steps. Cyclopropanation with
dimethylsulfoxonium methylide in DMSO gave desired trans cyclopropane
255 in �40% yield and 92% ee after recrystallization.
Hydrolysis of the ester followed by reaction with thionyl chloride
gave acid chloride 257 in 61% overall yield in two steps. Acid chloride 257 was then reacted with sodium azide in the presence
of sodium carbonate and tetrabutyl ammonium bromide in a biphasic
mixture of toluene and water to give the acyl azide intermediate,
which was immediately subjected to warm toluene to
furnish, after acidic workup, the key intermediate cyclopropyl
amine 237 in 88% yield and 92% ee. This enantioenriched intermediate was then mixed with R-(-)-mandelic acid to provide the
mandelic acid salt of amine 237 (258).

With all three intermediates available from the above mentioned
routes, the final assembly of ticagrelor was accomplished
as outlined in the scheme below. First, oxalate salt of cyclopentyl amine
235 was coupled with dichloroaminopyrimidine thioether 236 in the presence of triethylamine and at elevated temperature to give
diamine intermediate 259 in 88% yield after crystallization. Diamine
259 was then subjected to diazotization with sodium nitrite
in acetic acid and toluene at ??30??C, leading to the formation of triazole
260. This intermediate was immediately reacted with 258
(madelic acid salt of cyclopropyl amine 237) to give intermediate
261, which was subsequently taken forward to the final deprotection
step. Reaction of ketal 261 with concentrated HCl in methanol
and toluene at 15??C provided ticagrelor (XXII) in 82¨C90% yield
over the 3 steps. | | Drug interactions | Potentially hazardous interactions with other drugs
Antibacterials: concentration possibly increased by clarithromycin - avoid; concentration possibly increased by erythromycin; concentration reduced by rifampicin.
Anticoagulants: concentration of dabigatran increased.
Antifungals: concentration increased by ketoconazole - avoid.
Antivirals: concentration possibly increased by atazanavir and ritonavir - avoid.
Cardiac glycosides: concentration of digoxin increased.
Ciclosporin: possibly increases ciclosporin concentration.
Ergot alkaloids: concentration of ergot alkaloids possibly increased.
Lipid-regulating drugs: concentration of simvastatin increased - increased risk of toxicity. | | Metabolism | CYP3A4 is the major enzyme responsible for ticagrelor metabolism and the formation of the active metabolite and their interactions with other CYP3A substrates ranges from activation through to inhibition. The systemic exposure to the active metabolite is approximately 30-40
% of that obtained for ticagrelor. The primary route of ticagrelor elimination is via hepatic metabolism. The primary route of elimination for the active metabolite is most likely via biliary secretion. | | references | [1] zhou d1, andersson tb, grimm sw. in vitro evaluation of potential drug-drug interactions with ticagrelor: cytochrome p450 reaction phenotyping, inhibition, induction, and differential kinetics. drug metab dispos. 2011 apr;39(4):703-10.
[2] li y1, landqvist c, grimm sw. disposition and metabolism of ticagrelor, a novel p2y12 receptor antagonist, in mice, rats, and marmosets. drug metab dispos. 2011 sep;39(9):1555-67. doi: 10.1124/dmd.111.039669. epub 2011 jun 13. |
| | TICAGRELOR Preparation Products And Raw materials |
| Preparation Products | 2-[[(3aS,4R,6S,6aR)-4-[7-[[(1R,2S)-2-(3,4-Difluorophenyl)cyclopropyl]amino]-5-(propylthio)-3H-[1,2,3]triazolo[4,5-d]pyrimidin-3-yl]-2,2-dimethyl-tetrahydro-3aH-cyclopenta[d][1,3]dioxol-6-yl]oxy]ethanol |
|