- Imidazole
-

- $0.00 / 25KG
-
2024-04-26
- CAS:288-32-4
- Min. Order: 1KG
- Purity: 99%
- Supply Ability: 1000mt/year
- Imidazole
-

- $6000.00 / 1000kg
-
2024-04-25
- CAS:288-32-4
- Min. Order: 1000kg
- Purity: 99%
- Supply Ability: 500 tons
- Imidazole
-

- $6000.00 / 1000kg
-
2024-04-25
- CAS:288-32-4
- Min. Order: 1000kg
- Purity: 99%
- Supply Ability: 500 tons
Related articles - General Description of Imidazole
- Imidazole is a planar, five-membered, unsaturated, 6π electron ring system, comprised of three carbon atoms and two nitrogen ....
- Feb 11,2022
|
| | Imidazole Basic information |
| | Imidazole Chemical Properties |
| Melting point | 88-91 °C(lit.) | | Boiling point | 256 °C(lit.) | | density | 1.01 g/mL at 20 °C | | vapor pressure | <1 mm Hg ( 20 °C) | | refractive index | 1.4801 | | Fp | 293 °F | | storage temp. | Store below +30°C. | | solubility | H2O: 0.1 M at 20 °C, clear, colorless | | form | crystalline | | pka | 6.953(at 25℃) | | Specific Gravity | 1.03 | | color | white | | Odor | Amine like | | PH Range | 9.5 - 11 | | PH | 9.5-11.0 (25℃, 50mg/mL in H2O) | | Water Solubility | 633 g/L (20 ºC) | | Sensitive | Hygroscopic | | λmax | λ: 260 nm Amax: 0.10
λ: 280 nm Amax: 0.10 | | Merck | 14,4912 | | BRN | 103853 | | Dielectric constant | 23.0(Ambient) | | Stability: | Stable. Incompatible with acids, strong oxidizing agents. Protect from moisture. | | InChIKey | RAXXELZNTBOGNW-UHFFFAOYSA-N | | LogP | -0.02 at 25℃ | | CAS DataBase Reference | 288-32-4(CAS DataBase Reference) | | NIST Chemistry Reference | 1H-Imidazole(288-32-4) | | EPA Substance Registry System | Imidazole (288-32-4) |
| | Imidazole Usage And Synthesis |
| Description | Imidazole group denotes a heterocyclic organic compound whose molecule contains a five-membered hetero-aromatic ring of two non-adjacent nitrogen atoms, that a carbon atom is placed between two nitrogen atoms. Imidazole is the universally used trivial name for 1,3-azole. Earlier given names were glyoxaline and iminazole. The importance of this aromatic ring system is reflected by its presence in naturally occurring histidine, histamine and the purines, and in several classes of pharmaceuticals. Imidazole can act as a base and as a weak acid. It exists in two tautomeric forms with the hydrogen atom taking position between the two nitrogen atoms. | | Chemical Properties | Imidazole is a moderately strong base (pKb= 7.0), and a weak acid (pKa= 14.9). Imidazoles substituted with electron-withdrawing groups are stronger acids than imidazole itself; e.g., 4(5)-nitroimidazole has a pKa of 9.3. Imidazole is stable at 400°C, possesses considerable aromatic character, and undergoes the usual electrophilic aromatic substitution reactions. Nitration and sulfonation require, however, far more drastic conditions than the corresponding reactions with benzene. Other substitution reactions of imidazole include halogenation, hydroxymethylation, coupling with aromatic diazonium salts, and carboxylation. | | History | Imidazole[288-32-4] was first synthesized in 1858 by Debus from ammo�nia and glyoxal; it was originally named gly�oxalin. The name imidazole was introduced by Hantzsch. Industrial production of imidazole began in the 1950s; a wide range of derivatives is now available in industrial quantities. | | Uses | Imidazole is used as a buffer in the range of pH 6.2-7.8. It is also an histamine antagonist. It acts as a chelator and forms complexes with various divalent cations. It is used as a corrosion inhibitor on certain transition metals such as copper. Its derivatives, like polybenzimidazole (PBI), act as fire retardants. It finds application in photography and electronics. Imidazole salts are used as ionic liquids and precursors to stable carbenes. Imidazole derivatives like ketoconazole, miconazole and clotrimazole are involved in the treatment of various systemic fungal infections. It is a part of the theophylline molecule, present in tea leaves and coffee beans, which stimulates the central nervous system. | | Definition | ChEBI: Imidazole is an imidazole tautomer which has the migrating hydrogen at position 1. It is a conjugate base of an imidazolium cation. It is a conjugate acid of an imidazolide. It is a tautomer of a 4H-imidazole. It is an important pharmacophore in drug discovery. Imidazole is used as a Karl Fischer reagent in analytical chemistry and a reagent in synthetic organic chemistry. | | Application | Imidazole is a versatile heterocycle used in the preparation of various biologically active compounds such as the amino acid histidine and is present in many antifungal medication. It is also used ext ensively as a corrosion inhibitor on transition metals such as copper.It is used in organic synthesis and as an antiirradiationagent.
Imidazole has been used:
in the lysis, wash and elution buffer for the purification of histidine tagged Sonic Hedgehog(shh-N) protein.
in elution buffer in stepwise gradient for the purification of histidine tagged aldo keto reductases using nickel affinity chromatography.
as a component of homogenization buffer for the purification of phagosomal compartments from dendritic cell. | | Preparation | Imidazole is formed by reacting glyoxal with formaldehyde in the presence of ammonium acetate in acetic acid. The driving energy is microwave radiation. More generally, this reaction is used to produce substituted imidazoles.
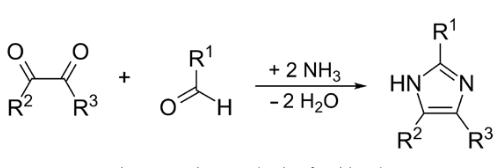
Although there had been discoveries of various derivatives of imidazole in 1840, it was first reported in 1858. The synthesis process of imidazole follows the reaction between formaldehyde in ammonia and glyoxal. This process gives low yield of imidazole but it is still used to form imidazole with C-substitution (Wolkenberg et al., 2004). | | General Description | Imidazole is a heterocyclic compound with a five-membered planar ring. It is amphoteric and highly polar. The pharmacophore of imidazole exists in bioactive compounds including amino acids, plant growth regulators and therapeutic agents. | | Health Hazard | It is less toxic relative to pyrrole and otherfive-membered heterocyclic compounds ofnitrogen. Intraperitoneal administration ofimidazole caused somnolence, muscle contractions,and convulsions in mice. Theoral LD50 value in mice is in the range900 mg/kg. | | Fire Hazard | Noncombustible solid. | | Biochem/physiol Actions | Imidazole derivatives have antibacterial, antifungal and anticancer functionality. It interacts with DNA and also binds to protein and stops cell division. It also acts as a microtubule destabilizing agents and inhibits topoisomerase and Cytochrome P450 Family 26 Subfamily A Member 1 (CYP26A1) enzymes. Imidazole based anticancer drug find applications in cancer chemotherapy. It is used as buffer component for purification of the histidine tagged recombinant proteins in immobilized metal-affinity chromatography (IMAC). | | Mechanism of action | N-substitution of imidazoles has created a family of drugs, called triazoles, that have the same mechanism of action as imidazoles, a similar or broader spectrum of activity, and less effect on human sterol synthesis. Both imidazoles and triazoles inhibit C-14α demethylation of lanosterol in fungi by binding to one of the cytochrome P-450 enzymes, which leads to the accumulation of C-14α methylsterols and reduced concentrations of ergosterol, a sterol essential for a normal fungal cytoplasmic membrane. Inhibition of cytochrome P-450 also decreases the synthesis of testosterone and glucocorticoids in mammals, an effect seen clinically with ketoconazole but not with later azoles[1]. | | Purification Methods | Crystallise imidazole from *benzene, CCl4, CH2Cl2, EtOH, pet ether, acetone/pet ether and distilled de-ionized water. Dry it at 40o under vacuum over P2O5. Distil it at low pressure. It is also purified by sublimation or by zone melting. [Snyder et al. Org Synth Coll Vol III 471 1955, Bredereck et al. Chem Ber 97 827 1964, Caswell & Spiro J Am Chem Soc 108 6470 1986.] 15N-imidazole crystallises from *benzene [Scholes et al. J Am Chem Soc 108 1660 1986]. [Beilstein 23 II 34, 23 III/IV 564, 23/4 V 191.] | | References | [1] Rex, J. and D. Stevens. “39 – Drugs Active against Fungi, Pneumocystis, and Microsporidia.” Mandell, Douglas, and Bennett’s Principles and Practice of Infectious Diseases 23 1 (2015): 479-494. |
| | Imidazole Preparation Products And Raw materials |
|