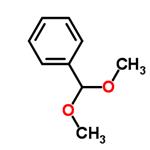
- Benzaldehyde dimethyl acetal
-
- $10.00 / 1KG
-
2024-01-14
- CAS:1125-88-8
- Min. Order: 1KG
- Purity: 99%
- Supply Ability: g-kg-tons, free sample is available
|
| | Benzaldehyde dimethyl acetal Basic information |
| | Benzaldehyde dimethyl acetal Chemical Properties |
| Hazard Codes | Xn | | Risk Statements | 22-36/37/38 | | Safety Statements | 23-24/25 | | RTECS | CU5774000 | | F | 10-21 | | TSCA | Yes | | HazardClass | IRRITANT | | HS Code | 29110000 | | Toxicity | LD50 orally in Rabbit: 1220 mg/kg LD50 dermal Rabbit > 5000 mg/kg |
| | Benzaldehyde dimethyl acetal Usage And Synthesis |
| Chemical Properties | Clear colorless liquid | | Chemical Properties | Benzaldehyde dimethyl acetal has a sweet, floral note; also reminiscent of almond | | Occurrence | Reported found in rhubarb and potato. | | Uses | Benzaldehyde dimethyl acetal may be used as an analytical reference standard for the quantification of the analyte in fresh and canned fish and Cinnamomum zeylanicum using gas-chromatography coupled to mass spectrometry (GC-MS). | | Uses | Benzaldehyde dimethyl acetal is suitable for use in the synthesis of 4,6-dihydroxy sugar, required for the total synthesis of Porphyromonas gingivalis 381 derived lipid A. It may be used in the preparation of 1-O-methyl-2,3-di-O-galloyl-β-D-glucose. | | Uses | Benzaldehyde dimethyl acetal used as an effective reagent for the construction of selenocarbonyl compounds. | | Definition | ChEBI: (Dimethoxymethyl)benzene is a benzyl ether. | | Preparation | From benzaldehyde and methanol in the presence of calcium chloride and HCl; from benzaldehyde and tetramethylorthosilicate in the presence of anhydrous HCl in methanol | | Preparation | A mixture of ethylenediamine (1.2 g, 20 mmol), Ph3SbO (1.0 mmol), and P4S10 (2.0 mmol) was autoclaved under a pressure of CO2 (4.9 MPa). Imidazolidinone was isolated by column chromatography (silica gel; eluent: ethyl acetate/hexane, 1:1, v/v); yield 1.5 g (85%). | | Taste threshold values | Taste characteristics at 25 ppm: green, nutty, brown and sweet | | General Description | The abstraction of α-hydrogen atoms from benzaldehyde dimethyl acetal by active bromine at 80°C has been investigated. | | Safety Profile | Moderately toxic by
ingestion. A skin irritant. When heated to
decomposition it emits acrid smoke and
irritating fumes. |
| | Benzaldehyde dimethyl acetal Preparation Products And Raw materials |
|